Research Article - Imaging in Medicine (2016) Volume 8, Issue 1
Diagnostic imaging and workup of malignant pleural mesothelioma
Edoardo Piacibello*Department of Diagnostic Imaging, San Luigi Gonzaga University Hospital, Orbassano, Torino, Italy
- Corresponding Author:
- Edoardo Piacibello
Department of Diagnostic Imaging
San Luigi Gonzaga University Hospital
Orbassano, Torino, Italy
Tel: 61 2 9736 1040
E-mail: dodo1986@hotmail.com
Abstract
Malignant pleural mesothelioma (MPM) is the most frequent primary neoplasm of the pleura. Although asbestos use has been banned in many developed countries, the incidence has been significantly increasing because of widespread occupational exposure over the last decades. Since the latency between first asbestos exposure and tumor development is around 40 years, the peak age incidence ranges from the sixth to the eighth decades and, since most asbestos exposure is work-related, the incidence is markedly higher in men than in women, the annual rates being 15 cases per million and 3 cases per million, respectively, in the United States. Most commonly, MPM originates within the parietal pleura located in the lower hemithorax and the costophrenic angle. It spreads locally to the ipsilateral visceral pleura and relentlessly invades adjacent structures, such as the lung, chest wall, diaphragm, pericardium, and mediastinum. Disease may invade the contralateral pleural space and the peritoneum. Lymphatic and hematogenous metastases tend to occur late in natural history but are present at autopsy in approximately 50% of patients with MPM. The clinical manifestations are nonspecific and many patients present with advanced-stage disease and comorbidities. The patient prognosis is poor, with a median survival after diagnosis of approximately 12 months. The diagnosis of this neoplasm is often made at a late stage and the prognosis is still very poor with a median survival from diagnosis of under a year with supportive care alone. Achieving early diagnosis and helping to select the most appropriate treatment option in MPM patients is mandatory. In this pictorial essay, the spectrum of imaging features of MPM at Chest Radiography (CXR), Computed Tomography (CT), Magnetic Resonance (MR), Positron Emission Tomography (PET), integrated PET/CT, and Ultrasonography (US) are discussed, and a diagnostic pathway in patients with undiagnosed pleural effusion is proposed.
Keywords
ultrasound; dorsal interosseous ligament; sacroiliac joint; sacroiliac joint incompetence; SPECT/CT
Introduction
The lifetime prevalence of low back pain has been reported to be as high as 84% with the prevalence of chronic low back pain being approximately 23% [1]. Between 11-12% of the population will be disabled by chronic low back pain [2]. Non-specific low back pain (NSLBP) is thought to be responsible for 85% of all cases of low back pain with approximately 15% being due to intervertebral disc disease and other causes [2,3]. Thus the vast majority of cases of low back pain will have no identifiable cause. This is supported by the literature which indicates that routine imaging of the lumbar spine utilising plain x-ray, CT or MRI does not add significantly to either functional or pain outcomes in patients with acute or subacute lower back pain [4].
The northern European literature has identified that approximately 15 to 21% of NSLBP may be due to mechanical dysfunction of the sacroiliac joint (SIJ) [5-7]. Prevalence of this diagnosis has been common enough for publication of evidence-based guidelines for the diagnosis and management of such patients [8]. The importance of identification of these cases is that with specific therapy, approximately 80% will regain good functional and pain outcomes [9,10]. A proportion that does not respond to physiotherapy appears to respond to imageguided prolotherapy with hypertonic dextrose [9]. We hypothesised that ultrasound-guided injection into the posterior sacroiliac joint ligamentous complex could provide a similar response while avoiding the radiation exposure from either x-ray computed tomography or screening. Reproduction of clinical symptoms by entry of the needle into the dorsal interosseous ligament was utilised as a reference standard for the procedure as part of a prospective outcomedriven trial.
Imaging
CRX is usually the first-line radiologic examination, but the radiographic findings are nonspecific and others imaging modalities such as CT, MR, PET-CT and US are indicated.
CT is the mainstay imaging technique for primary assessment of pleural disease and affords improved sensitivity and specificity for identification of malignant pleural process. MRI, PET or PET/CT and US are complementary techniques for the assessment of pleural disease that can provide additional important diagnostic, staging and prognostic information.
Plain chest radiography (CXR)
CXR, due to its ready availability, is usually the first imaging modality used to detect abnormalities
suggesting MPM. The radiographic appearance of MPM is variable and depends on the stage of disease at diagnosis.
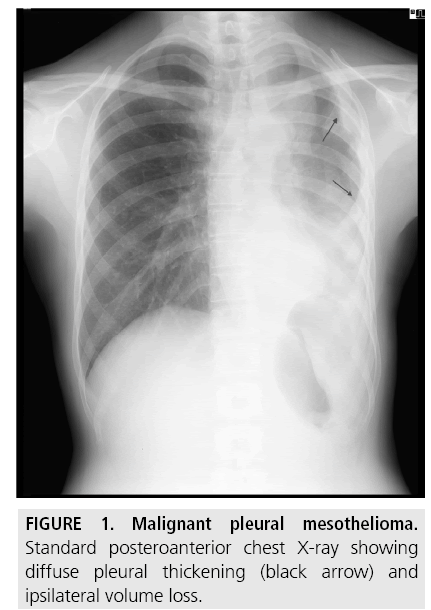
A unilateral pleural effusion is the typical finding at presentation and is seen in 30%-80% of patients (FIGURE 1).
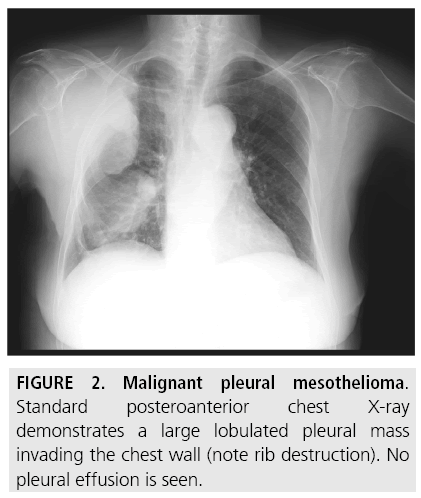
A pleural-based mass, in the absence of pleural effusion, is shown in less than 25% of patients [6] (FIGURE 2). Diffuse pleural thickening or extensive lobular pleural-based masses are seen in about half of cases [6].
Tumor growth leads to nodular thickening of interlobar fissures and lung encasement with a rind-like appearance, ipsilateral volume loss and mediastinal shift. Larger pleural-based masses often coexist with multiloculated effusions which tend to obscure the underlying neoplasm [7].
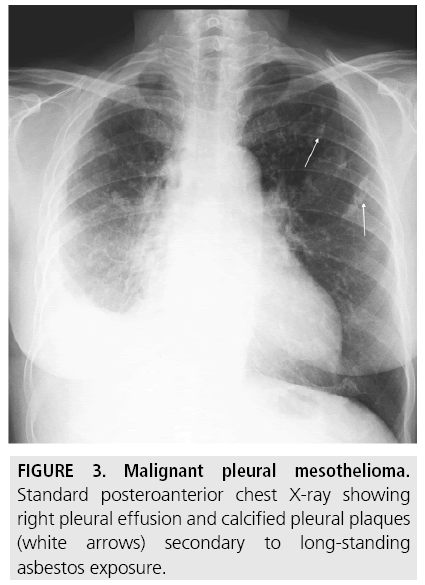
Pleura plaques are thickened areas of parietal pleura composed of connective tissue which can undergo calcification, and are probably the commonest radiographic manifestation of long-standing asbestos exposure, seen in approximately 20% of cases (FIGURE 3). They are more prominent on the domes of the diaphragm and in the lower half of the thorax. Combined pleural and parenchymal changes can cause the “shaggy” heart sign, a partial obscuration of the heart border [1,7]. Although the presence of pleural plaques alone does not per se require additional diagnostic workup, a statistically significant association was observed in a 7-year follow-up study of formerly asbestosexposed workers between pleural plaques, detected on CT, and the risk of MPM [8].
Computed Tomography (CT)
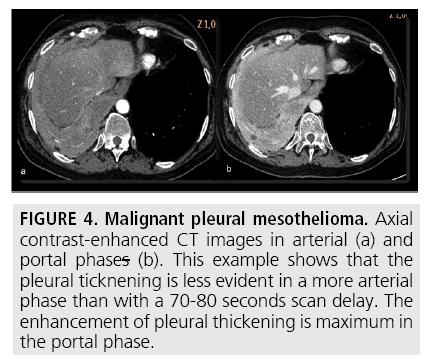
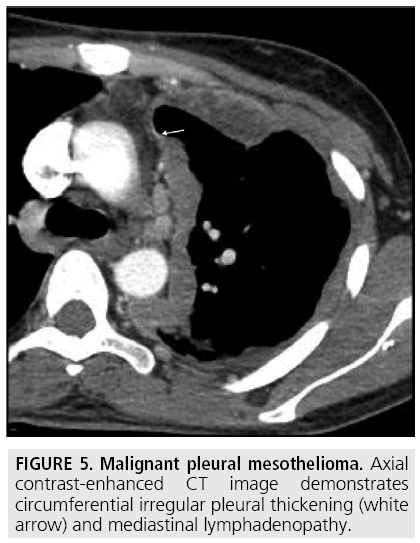
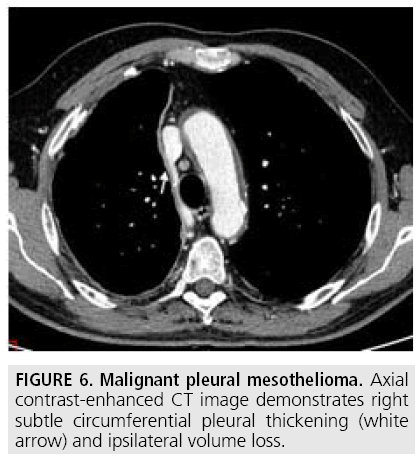
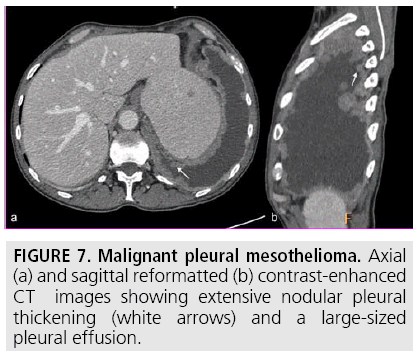
Computed tomography (CT) continues to be the mainstay imaging technique for the initial assessment of MPM and plays a primary role in structuring the subsequent diagnostic and staging evaluation as well as therapeutic decision-making process. Technical CT factors are very important for reaching the correct diagnosis. The last generation CT technology (> 32 detector rows) allows thin-section volumetric acquisitions providing an isotropic data set, which can be reconstructed in any plane. As a result, these multiplanar reformations allow to easily evaluating the presence of very limited pleural thickening. Employment of a contrast medium is mandatory [1], the CT scanning delay should be also set at 60-80 seconds to optimize the maximum pleural tumor uptake [3] (FIGURE 4) and the field-of- view (FOV) due to the tumor growth through the diaphragmatic pillars had to cover a wide area from the lung apex to the to L3. CT features highly suggestive of the disease include nodular or lobular pleural thickening, interlobar fissure thickening, mediastinal pleural thickening, parietal pleural thickening > 1 cm, and circumferential pleural thickening. The most common CT finding is pleural thickening and is seen in 90%- 92% of patients [9]. It greatly varies in extent, thickness, and nodularity. Circumferential pleural thickening with rind-like encasement of the lung and ipsilateral volume loss is seen in advanced-stage disease. Focal pleural masses of > 3 cm in diameter are identified in 8%-38% of cases. The next most frequent CT finding is interlobar fissure involvement and is identified, as thickening and/or nodularity, in 73%-86% of patients. Additional CT findings include pleural effusions and plaques and are seen in approximately 75% and 20% of cases, respectively.
Figure 4: Malignant pleural mesothelioma. Axial contrast-enhanced CT images in arterial (a) and portal phases (b). This example shows that the pleural ticknening is less evident in a more arterial phase than with a 70-80 seconds scan delay. The enhancement of pleural thickening is maximum in the portal phase.
MPM has a propensity for early invasion into adjacent structures. Mediastinal pleura, vascular structures and organs involvement may result in obliteration of fat planes and encasement of great vessels, esophagus and trachea. Involvement of the pericardium can be seen as pericardial thickening and/or effusion. Extension of the tumor into the chest wall may result in obliteration of extrapleural fat planes, invasion of intercostal muscles, and rib displacement or destruction. Thickening of the hemidiaphragm is a common finding. However, CT has shown poor/limited accuracy in identifying transdiaphragmatic tumor extension. Features suggesting transdiaphragmatic invasion include a soft tissue mass that encases the hemidiaphragm and absence of a fat plane between the inferior surface of the muscle and adjacent abdominal organs. Finally, CT is useful for the evaluation of intrathoracic lymphadenopathy.
Over the last decades, a number of staging systems have been proposed to predict outcome and guide appropriate treatment planning in MPM patients. The International Mesothelioma Interest Group [10] developed a new staging system based on primary tumor local extent (T), lymph node involvement (N), and metastatic disease (M).
Accurate staging based on imaging is pivotal for identifying potential candidates to aggressive surgical procedures and multimodality treatment. However, CT has repeatedly shown limited accuracy in distinguishing between potentially resectable (T3) and technically unresectable (T4) disease as well as in identifying intrathoracic lymph node involvement [11,12].
Finally, several authors have shown the value of CT in differentiating benign from malignant pleural disease [13-15]. Helpful discriminating features of malignant disease on CT scanning include nodular pleural thickening, mediastinal pleural thickening, parietal pleural thickening > 1 cm, and circumferential pleural thickening. However, data from a recent study [16] suggest that although the sensitivity of these findings is higher than previously reported (68%), the specificity is significantly lower (78%). Of note, with a negative predictive value of 65%, the absence of these findings does not exclude malignant pleural disease. Besides, these findings have shown a limited importance for differentiation of MPM from metastatic pleural disease [14,15].
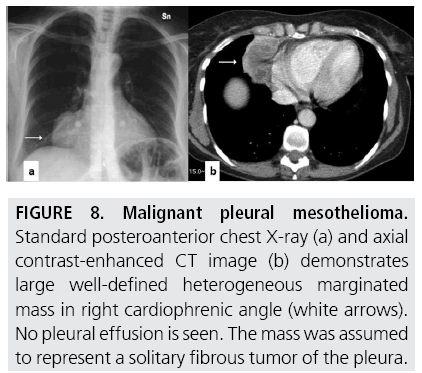
FIGURES 5 to 9 illustrate some of the findings typically seen on CT imaging in MPM.
Figure 8: Malignant pleural mesothelioma. Standard posteroanterior chest X-ray (a) and axial contrast-enhanced CT image (b) demonstrates large well-defined heterogeneous marginated mass in right cardiophrenic angle (white arrows). No pleural effusion is seen. The mass was assumed to represent a solitary fibrous tumor of the pleura.
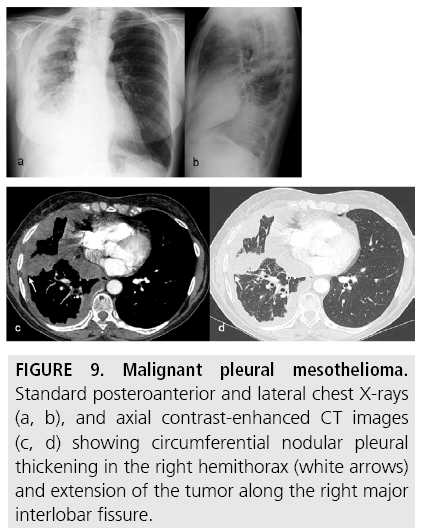
Figure 9: Malignant pleural mesothelioma. Standard posteroanterior and lateral chest X-rays (a, b), and axial contrast-enhanced CT images (c, d) showing circumferential nodular pleural thickening in the right hemithorax (white arrows) and extension of the tumor along the right major interlobar fissure.
Magnetic Resonance (MR)
Because of cost reasons, limited availability, and long imaging time, MR is not commonly used in the diagnostic and staging evaluation of MPM patients. However, owing to excellent contrast resolution on unenhanced scans and higher enhancement achieved post-contrast, it has been found useful in equivocal cases as well as in potential candidates to multimodality therapy including surgery [17-19]. Indeed, the combination of morphological data and information on signal intensity may provide more precise assessment of local disease extent [19].
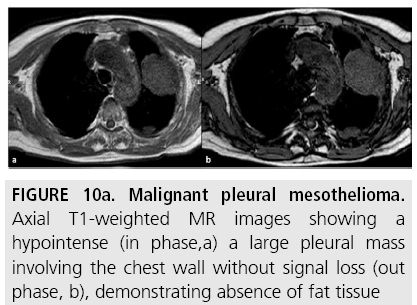
Pleural mesothelioma is characterized by intermediate or slightly hyperintense signal on T1-weighted sequences (FIGURE 10a) and by more intense signal on T2-weighted sequences, compared with adjacent chest wall healthy tissue (FIGURE 10b) [20]. The signal of pleural mesothelioma may be further enhanced by using gadolinium-based paramagnetic contrast material. Contrast-enhanced T2-weighted fat suppressed sequences (FIGURE 10b) are the most sensitive sequences for detecting invasion of interlobar fissures and of adjacent structures [17]. Furthermore, diffusion-weighted MR (FIGURE 10c) can reveal tissue characteristics based on the diffusivity of water molecules within tissues. With this technique, signal loss can be quantitatively assessed with the apparent diffusion coefficient (ADC), which depends on restriction of water molecules diffusion by cell membranes and macromolecules, indirectly providing information about tissue cellularity [18]. As for the assessment of local disease extent, Patz and colleagues [11] compared MR with CT in 34 MPM patients undergoing thoracotomy. Review of imaging findings focused on local invasion of the diaphragm, chest wall, and mediastinum. MR showed slightly higher sensitivity than CT for predicting respectability at the diaphragm and chest wall (100% vs. 93%-94%, respectively), most likely because MR provided additional coronal and sagittal images. Heelan and colleagues [17] compared the accuracy of MR with that of CT in the preoperative staging of 65 MPM patients. MR and CT imaging showed nearly equivalent diagnostic accuracy in staging, but MR was more accurate for detecting solitary foci of chest wall invasion and endothoracic fascia involvement and for assessing diaphragmatic invasion. However, these findings did not change the surgical approach. Furthermore, the higher resolution and the ability for multiplanar reformations afforded by multidetector CT (MDCT) may provide more accurate assessment of the local extent of MPM.
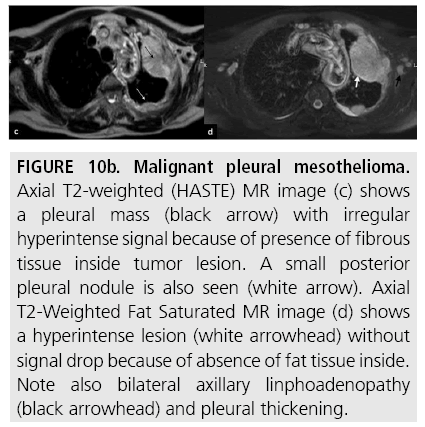
Figure 10b: Malignant pleural mesothelioma. Axial T2-weighted (HASTE) MR image (c) shows a pleural mass (black arrow) with irregular hyperintense signal because of presence of fibrous tissue inside tumor lesion. A small posterior pleural nodule is also seen (white arrow). Axial T2-Weighted Fat Saturated MR image (d) shows a hyperintense lesion (white arrowhead) without signal drop because of absence of fat tissue inside. Note also bilateral axillary linphoadenopathy (black arrowhead) and pleural thickening.
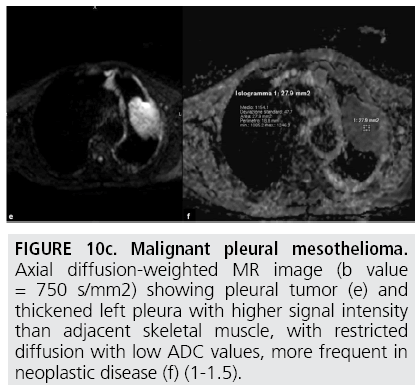
Figure 10c: Malignant pleural mesothelioma. Axial diffusion-weighted MR image (b value = 750 s/mm2) showing pleural tumor (e) and thickened left pleura with higher signal intensity than adjacent skeletal muscle, with restricted diffusion with low ADC values, more frequent in neoplastic disease (f) (1-1.5).
Positron-Emission Tomography (PETCT)
Owing to the ability of providing both metabolic and anatomic information about a lesion, PET and PET/CT have emerged as important complementary techniques for the assessment of pleural disease.
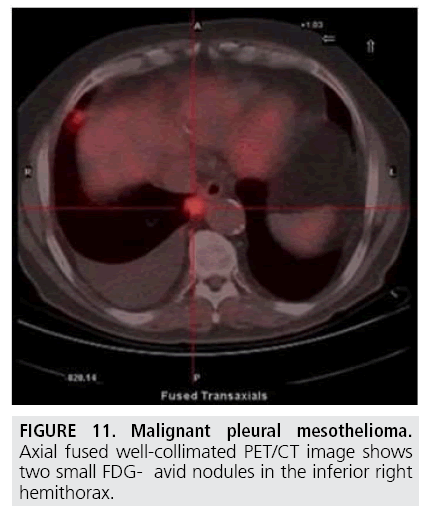
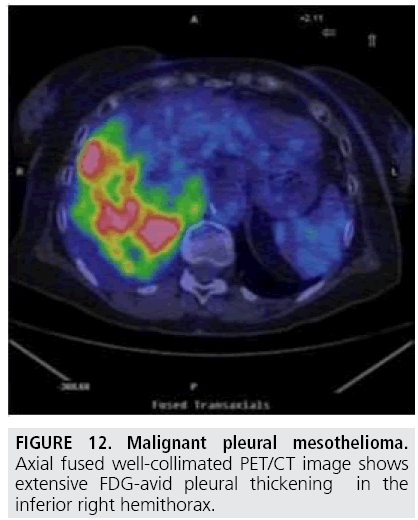
The elevated metabolic activity of tumor cells results in significantly higher 18F-Fluorodeoxyglucose standardized uptake value (SUV) of MPM compared with benign pleural diseases. Several authors [21-25] showed that a SUV cutoff value of 2.0-2.2 differentiated malignant from benign pleural disease with sensitivities of 91%-100% and specificities of 78%-100% (FIGURE 11). In addition, PET has been found useful to identify the most appropriate biopsy site for achieving definite diagnosis (FIGURE 12). However, PET accuracy in distinguishing benign and malignant pleural disease is limited by false-negative (low-grade variant of MPM) and false-positive [26] (concomitant asbestos-related disease, parapneumonic effusion, uraemic pleural disease, and talc pleurodesis) results while PET has demonstrated suboptimal sensitivity and specificity in staging MPM patients [27-29].
Due to superior anatomic spatial resolution, integrated PET/CT has been increasingly used for diagnostic and staging evaluation as well as treatment planning of MPM. PET/CT has demonstrated better accuracy in overall staging of MPM patients and in identifying potential candidates to multimodality therapy including aggressive surgical procedures. Indeed, two reviews evaluated the staging information of PET/TC and showed a wide range of accuracy for T, N, and M descriptors [30,31]. Recently, Frauenfelder and colleagues [12] evaluated the accuracy of CT and PET/CT for MPM staging in 28 patients undergoing induction chemotherapy. CT and PET/ CT underestimated T stage in up to 30% of patients. PET/CT showed higher accuracy for tumor extent compared with CT (92% vs. 84%, respectively) while CT showed higher accuracy for N staging compared with PET/CT (87% vs. 78%, respectively). Regarding the International Mesothelioma Interest Group staging system [10], the accuracy of PET/CT in preoperative staging was higher compared with CT (91% vs. 82%, respectively). Furthermore, the interobserver agreement for local tumor extent and N staging was lower for CT compared with PET/CT.
PET/CT may also have a role for monitoring treatment response, detecting recurrent disease, and providing prognostic information in MPM patients [29,31].
Ultrasonography (US)
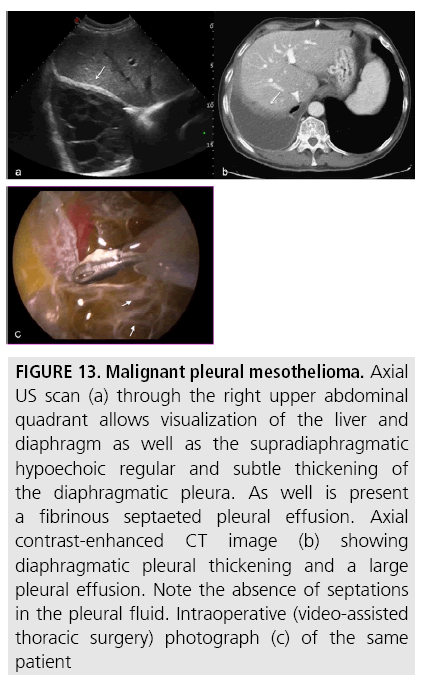
In the initial evaluation of pleural effusions, US has shown high sensitivity in pleural fluid detection and quantification [32,33]. It plays a pivotal role in image-guided techniques (thoracocentesis, needle biopsy, drain placement) and identifies complex, septated patterns of pleural effusion with higher sensitivity than CT (FIGURE 13).
Figure 13: Malignant pleural mesothelioma. Axial US scan (a) through the right upper abdominal quadrant allows visualization of the liver and diaphragm as well as the supradiaphragmatic hypoechoic regular and subtle thickening of the diaphragmatic pleura. As well is present a fibrinous septaeted pleural effusion. Axial contrast-enhanced CT image (b) showing diaphragmatic pleural thickening and a large pleural effusion. Note the absence of septations in the pleural fluid. Intraoperative (video-assisted thoracic surgery) photograph (c) of the same patient
Pleural thickening most often appears hypoechoic, but increased echogenicity with focal acustic shadowing is seen in presence of calcification and fibrosis (pleural plaques) [34-38]. Mesotheliomas have very irregular, partly angular, unclear borders. In addition to tumorlike formations, mesotheliomas can also present as extensive, tapestry- like growths with nodules. Using high-frequency transducers, invasions of the chest wall and the diaphragm are visualized as striped, hypoechoic ramifications at the time of diagnosis [39].
By using similar morphologic criteria as those used in CT (pleural thickening >1 cm, pleural and diaphragmatic thickening >7 mm), Qureshi and colleagues [40] demonstrated that US is able to differentiate malignant from benign effusions with an overall sensitivity of 79% and specificity of 100%, with specificity comparing favourably with CT. The authors’ conclusions were that US, being a quick, relatively inexpensive and harmless procedure, may represent a valuable adjunct in the diagnostic pathway of suspected malignant pleural effusion.
Diagnostic pathway: our experience
We participated and contributed to the 2nd Italian Consensus Conference on Malignant Pleural Mesothelioma [41] held in Turin (Italy) on November 24-25, 2011. In the light of this starting point and of several international guidelines [42-45], we adopted a tailored diagnostic pathway, based on our experience and hospital facilities, as much as rational and cost-effective as possible in a high-risk area.
The chest X-ray (CXR) remains the first imaging modality for the approach to patients with suspected MPM. The CXR finding of pleural plaques does not require additional investigations [42], whereas recurrent unilateral pleural effusion [43] not related to any known etiology such as infection or congestive heart failure should be further investigated by CT with contrast medium.
According to the MDCT findings, our targeted diagnostic workup may be summarized as follows:
1. In patients presenting with dyspnea due to a pleural effusion, if the clinician has any suspicion for a malignancy, a US guided thoracentesis should be performed.
2. Presence of gross irregular pleural masses (with or without pleural effusion) should be further investigated by US or CT guided-biopsy.
3. A limited irregular pleural thickening (with or without pleural effusion) may be evaluated by PET-CT scanning.
4. Recurrent pleural effusion without any visible abnormality at CT scan should be directly investigated by video assisted thoracoscopy (VATS), a minimally invasive technique with a high diagnostic yield which allows exploration of entire pleural surface and enables targeted biopsies, providing material samples for both histological examination and immune histochemical analysis.
5. MRI is used when there are contraindications to iodinated contrast medium and to provide more accurate assessment of chest wall or diaphragmatic invasion in patients deemed potential candidates to aggressive multimodality therapeutic regimens.
Conclusion
Imaging of MPM is a challenge for the radiologist because the pleural surface has a complex shape and the disease shows an asymmetric growth and tendency to infiltrate locally along tissue planes.
Each imaging modality has its strengths and limitations, but their rational and cost-effective combined use is crucial in determining the most appropriate treatment options for patients with MPM.
References
- Nickell LT Jr, Lichtenberger JP , Khorashadi L et al. Multimodality imaging for characterization, classification, and staging of malignant pleural mesothelioma. Radiographics. 34: 1692-1706 (2014).
- Rusch VW. Diffuse malignant mesothelioma. In: Shields TW, LoCicero J III, Reed CE, Feins RH (Eds), General thoracic surgery. Wolters Kluwer/Lippincott Williams & Wilkins, Philadelphia (2009).
- Raj V, Kirke R, Bankart MJ et al. Multidetector CT imaging of pleura: comparison of two contrast infusion protocols. Br. J. Radiol. 84, 796-799 (2011).
- Boutin C, Rey F. Thoracoscopy in pleural malignant mesothelioma: a prospective study of 188 consecutive patients. Part 1: Diagnosis. Cancer. 72, 389-393 (1993).
- Flores RM. The impact of lymph node station on survival in 348 patients with surgically resected malignant pleural mesothelioma: implications for revision of the American Joint Committee on Cancer staging system. J. Thorac. Cardiovasc. Surg. 136, 605-610 (2008).
- Cardinale L, Ardissone F, Asteggiano F et al. Diffuse neoplasms of the pleural serosa. Radiol. Med. 118, 366-378 (2013).
- Peacock C, Copley SJ, Hansell DM. Asbestosrelated benign pleural disease. Clin. Radiol. 55, 422-432 (2000).
- Pairon JC, Laurent F, Rinaldo M et al. Pleural plaques and the risk of pleural mesothelioma. J. Natl. Cancer Inst. 105, 293-301 (2013).
- Tamer Dogan O, Salk I, Tas F et al. Thoracic computed tomography findings in malignant mesothelioma. Iran J. Radiol. 9, 209-211 (2012).
- Rusch VW. A proposed new international TNM staging system for malignant pleural mesothelioma. From the International Mesothelioma Interest Group. Chest. 108, 1122-1128 (1995).
- Patz EF Jr, Shaffer K, Piwnica-Worms DR et al. Malignant pleural mesothelioma: value of CT and MR imaging in predicting resectability. AJR Am. J. Roentgenol. 159, 961-966 (1992).
- Frauenfelder T, Kestenholz P, Veit-Haibach P et al. Use of computed tomography and positron emission tomography/computed tomography for staging of local extent in patients with malignant pleural mesothelioma. J. Comput. Assist. Tomogr. (2014).
- Maffessanti M, Tommasi M, Pellegrini P. Computed tomography of free pleural effusions. Eur. J. Radiol. 7, 87-90 (1987).
- Leung AN, Müller NL, Miller RR CT in differential diagnosis of diffuse pleural disease. AJR Am. J. Roentgenol. 154, 487-492.(1990).
- Metintas M. Computed tomography features in malignant pleural mesothelioma and other commonly seen pleural diseases. Eur. J. Radiol. 41(1), 1-9 (2012).
- Hallifax RJ, Haris M, Corcoran JP et al. Role of CT in assessing pleural malignancy prior to thoracoscopy. Thorax .70, 192-193 (2015).
- Heelan RT, Rusch VW, Begg CB et al. Staging of malignant pleural mesothelioma: comparison of CT and MR imaging. AJR Am. J. Roentgenol. 172, 1039-1047 (1999).
- Knuuttila A, Kivisaari L, Kivisaari A et al. Evaluation of pleural disease using MR and CT. With special reference to malignant pleural mesothelioma. Acta. Radiol. 42, 502-507 (2001).
- Gill RR, Umeoka S, Mamata H et al. Diffusion-weighted MRI of malignant pleural mesothelioma: preliminary assessment of apparent diffusion coefficient in histologic subtypes. Am. J. Roentgenol. 195, W125-30 (2010).
- Zucali PA, Giaccone G. Biology and management of malignant pleural mesothelioma. Eur. J. Cancer. 42, 2706-2714 (2006).
- Yildirim H, Metintas M, Entok E. Clinical value of fluorodeoxyglucose-positron emission tomography/computed tomography in differentiation of malignant mesothelioma from asbestosrelated benign pleural disease: an observational pilot study. J. Thorac. Oncol. 4, 1480-1484 (2009).
- Bénard F, Sterman D, Smith RJ et al. Metabolic imaging of malignant pleural mesothelioma with fluorodeoxyglucose positron emission tomography. Chest. 114, 713-722 (1998).
- Duysinx B, Nguyen D, Louis R et al. Evaluation of pleural disease with 18-fluorodeoxyglucose positron emission tomography imaging. Chest 125, 489-493 (2004).
- Kramer H, Pieterman RM, Slebos DJ et al. PET for the evaluation of pleural thickening observed on CT. J. Nucl. Med. 45, 995-998 (2004).
- Orki A, Akin O, Tasci AE et al. The role of positron emission tomography/computed tomography in the diagnosis of pleural diseases. Thorac. Cardiovasc. Surg. 57, 217-221 (2009).
- Treglia G, Sadeghi R. Diagnostic accuracy of 18F-FDG-PET and PET/CT in the differential diagnosis between malignant and benign pleural lesions: a systematic review and metaanalysis. Academic Radiol. 21, 11-20 (2014).
- Schneider DB, Clary-Macy C, Challa S. Positron emission tomography with F18- fluorodeoxyglucose in the staging and preoperative evaluation of malignant pleural mesothelioma. J. Thorac. Cardiovasc. Surg. 120, 128-133 (2000).
- Wang ZJ, Reddy GP, Gotway MB et al. Malignant pleural mesothelioma: evaluation with CT, MR imaging, and PET. Radiographics 24, 105-119 (2004).
- Flores RM. Positron emission tomography defines metastatic disease but not locoregional disease in patients with malignant pleural mesothelioma. J. Thorac. Cardiovasc. Surg. 126(1), 11-6 (2003).
- Basu S, Saboury B, Torigian DA. Current evidence base of FDG-PET/CT imaging in the clinical management of malignant pleural mesothelioma: emerging significance of image segmentation and global disease assessment. Mol. Imaging. Biol. 13(5), 801-11 (2011).
- Sharif S, Zahid I, Routledge T,et al. Does positron emission tomography offer prognostic information in malignant pleural mesothelioma? Interact. Cardiovasc. Thorac. Surg. 12, 806-811 (2011).
- Rahman NM, Davies RJ, Gleeson FV. Investigating suspected malignant pleural effusion. BMJ 334, 206-207 (2007).
- McLoud TC, Flower CD. Imaging the pleura: sonography, CT, and MR imaging. AJR Am. J. Roentgenol. 156, 1145-1153 (1991).
- Mathis G [Introduction--emergency ultrasonography]. Ultraschall. Med. 29, 451- 452 (2008).
- Görg C, Bert T, Görg K, et al. Colour Doppler ultrasound mapping of chest wall lesions. Br. J. Radiol. 78, 303-307 (2005).
- Saito T, Kobayashi H, Kitamura S. Ultrasonographic approach to diagnosing chest wall tumors. Chest. 94, 1271-1275 (1988).
- Bandi V, Lunn W, Ernst A et al. Ultrasound vs. CT in detecting chest wall invasion by tumor: a prospective study. Chest. 133, 881-886 (2008).
- Popic Ramac J. The possibilities and limitations of direct digital radiography, ultrasound and computed tomography in diagnosing pleural mesotelioma. Coll. Antropol. 34(4), 1263-71 (2010).
- Reuss. The Pleura in Gebhard Mathis (Ed.) Chest Sonography, SBN 978-3-540-72427-8 Springer Berlin Heidelberg New York . 36 -37.
- Qureshi NR, Rahman NM, Gleeson FV. Thoracic ultrasound in the diagnosis of malignant pleural effusion. Thorax. 64, 139- 143 (2009).
- Pinto C, Betta PG, Fava C et al. Second Italian consensus conference on malignant pleural mesothelioma: state of the art and recommendations. Cancer. Treat. Rev. 39, 328- 39 (2013).
- Armato SG, Labby ZE, Coolen J et al. Imaging in pleural mesothelioma: a review of the 11th International Conference of the International Mesothelioma Interest Group. Lung. Cancer. 82, 190-196 (2013).
- Hooper C, Lee YC, Maskell N. BTS Pleural Guideline Group. Investigation of a unilateral pleural effusion in adults: British Thoracic Society Pleural Disease Guideline. Thorax. 2, ii4-17 (2010).
- Stahel RA, Weder W, Lievens Y et al. ESMO Guidelines Working Group Malignant pleural mesothelioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and followup. Ann. Oncol. 21 126-128 (2010).
- van Zandwijk N, Clarke C, Henderson D et al. Guidelines for the diagnosis and treatment of malignant pleural mesothelioma. J. Thorac. Dis. 5, E254-307 (2013).
- Nickell LT Jr, Lichtenberger JP , Khorashadi L et al. Multimodality imaging for characterization, classification, and staging of malignant pleural mesothelioma. Radiographics. 34, 1692-1706 (2014).
- Rusch VW. Diffuse malignant mesothelioma. In: Shields TW, LoCicero J III, Reed CE, Feins RH (Eds), General thoracic surgery. Wolters Kluwer/Lippincott Williams & Wilkins, Philadelphia. 847–859 (2009).
- Raj V, Kirke R, Bankart MJ. Multidetector CT imaging of pleura: comparison of two contrast infusion protocols. Br. J. Radiol. 84, 796-799 (2011).
- Boutin C, Rey F. Thoracoscopy in pleural malignant mesothelioma: a prospective study of 188 consecutive patients. Part 1: Diagnosis. Cancer. 72, 389-393 (1993).
- Flores RM. The impact of lymph node station on survival in 348 patients with surgically resected alignant pleural mesothelioma: implications for revision of the American Joint Committee on Cancer staging system. J. Thorac. Cardiovasc. Surg. 136, 605-610 (2008).
- Cardinale L, Ardissone F, Asteggiano F et al. Diffuse neoplasms of the pleural serosa. Radiol. Med. 118, 366-378 (2013).
- Peacock C, Copley SJ, Hansell DM. Asbestosrelated benign pleural disease. Clin. Radiol. 55, 422-432 (2000).
- Pairon JC, Laurent F, Rinaldo M, et al. Pleural plaques and the risk of pleural mesothelioma. J. Natl. Cancer Inst. 105, 293-301 (2013).
- Tamer Dogan O, Salk I, Tas F, et al. Thoracic computed tomography findings in malignant mesothelioma. Iran. J. Radiol. 9, 209-211 (2012).
- Rusch VW. A proposed new international TNM staging system for malignant pleural mesothelioma. From the International Mesothelioma Interest Group. Chest. 108, 1122-1128 (1995).
- Patz EF Jr, Shaffer K, Piwnica-Worms DR et al. Malignant pleural mesothelioma: value of CT and MR imaging in predicting resectability. AJR Am. J. Roentgenol. 159, 961-966 (1992).
- Frauenfelder T, Kestenholz P, Veit-Haibach P et al. Use of computed tomography and positron emission tomography/computed tomography for staging of local extent in patients with malignant pleural mesothelioma. J. Comput. Assist. Tomogr. (2014).
- Maffessanti M, Tommasi M, Pellegrini P. Computed tomography of free pleural effusions. Eur. J. Radiol. 7, 87-90 (1987).
- Leung AN, Müller NL, Miller RR. CT in differential diagnosis of diffuse pleural disease. AJR Am. J. Roentgenol. 154, 487-492 (1990).
- Metintas M. Computed tomography features in malignant pleural mesothelioma and other commonly seen pleural diseases. Eur. J. Radiol. 41(1), 1-9 (2012).
- Hallifax RJ, Haris M, Corcoran JP et al. Role of CT in assessing pleural malignancy prior to thoracoscopy. Thorax 70, 192-193 (2015).
- Heelan RT, Rusch VW, Begg CB et al. Staging of malignant pleural mesothelioma: comparison of CT and MR imaging. AJR Am. J. Roentgenol. 172, 1039-1047 (1999).
- Knuuttila A, Kivisaari L, Kivisaari A et al. Evaluation of pleural disease using MR and CT. With special reference to malignant pleural mesothelioma. Acta. Radiol. 42, 502-507 (2001).
- Gill RR, Umeoka S. Diffusion-weighted MRI of malignant pleural mesothelioma: preliminary assessment of apparent diffusion coefficient in histologic subtypes.Am. J. Roentgenol. 195, W125-30 (2010).
- Zucali PA, Giaccone G. Biology and management of malignant pleural mesothelioma. Eur. J. Cancer. 42, 2706-2714 (2006).
- Yildirim H, Metintas M, Entok E. Clinical value of fluorodeoxyglucose-positron emission tomography/computed tomography in differentiation of malignant mesothelioma from asbestosrelated benign pleural disease: an observational pilot study. J. Thorac. Oncol. 4, 1480-1484 (2009).
- Bénard F, Sterman D, Smith RJ, Metabolic imaging of malignant pleural mesothelioma with fluorodeoxyglucose positron emission tomography. Chest. 114, 713-722 (1998).
- Duysinx B, Nguyen D, Louis R et al. Evaluation of pleural disease with 18-fluorodeoxyglucose positron emission tomography imaging. Chest. 125, 489-493 (2004).
- Kramer H, Pieterman RM, Slebos DJ et al. PET for the evaluation of pleural thickening observed on CT. J. Nucl. Med. 45, 995-998 (2004).
- Orki A, Akin O, Tasci AE et al. The role of positron emission tomography/computed tomography in the diagnosis of pleural diseases. Thorac. Cardiovasc. Surg. 57, 217-221 (2009).
- Treglia G, Sadeghi R. Diagnostic accuracy of 18F-FDG-PET and PET/CT in the differential diagnosis between malignant and benign pleural lesions: a systematic review and metaanalysis. Academic. Radiol. 21, 11-20 (2014).
- Schneider DB, Clary-Macy C, Challa S. Positron emission tomography with F18- fluorodeoxyglucose in the staging and preoperative evaluation of malignant pleural mesothelioma. J. Thorac. Cardiovasc. Surg. 120, 128-133 (2000).
- Wang ZJ, Reddy GP, Gotway MB et al. Malignant pleural mesothelioma: evaluation with CT, MR imaging, and PET. Radiographics. 24, 105-119 (2004).
- Flores RM. Positron emission tomography defines metastatic disease but not locoregional disease in patients with malignant pleural mesothelioma. J. Thorac. Cardiovasc. Surg. 126(1), 11-6 (2003).
- Basu S, Saboury B, Torigian DA. Current evidence base of FDG-PET/CT imaging in the clinical management of malignant pleural mesothelioma: emerging significance of image segmentation and global disease assessment. Mol. Imaging. Biol. 13(5), 801-11 (2011).
- Sharif S, Zahid I, Routledge T et al. Does positron emission tomography offer prognostic information in malignant pleural mesothelioma? Interact. Cardiovasc. Thorac. Surg. 12, 806-811 (2011).
- Rahman NM, Davies RJ, Gleeson FV. Investigating suspected malignant pleural effusion. BMJ 334: 206-207 (2007).
- McLoud TC1, Flower CD. Imaging the pleura: sonography, CT, and MR imaging. AJR Am. J. Roentgenol. 156, 1145-1153 (1991).
- Mathis G. [Introduction--emergency ultrasonography]. Ultraschall. Med. 29, 451- 452 (2008).
- Görg C, Bert T, Görg K et al. Colour Doppler ultrasound mapping of chest wall lesions. Br. J. Radiol. 78, 303-307 (2005).
- Saito T, Kobayashi H, Kitamura S. Ultrasonographic approach to diagnosing chest wall tumors. Chest 94, 1271-1275 (1988).
- Bandi V, Lunn W, Ernst A et al. Ultrasound vs. CT in detecting chest wall invasion by tumor: a prospective study. Chest. 133, 881-886 (2008).
- Popic Ramac J. The possibilities and limitations of direct digital radiography, ultrasound and computed tomography in diagnosing pleural mesotelioma. Coll. Antropol. 34(4), 1263-71 (2010).
- Reuss. The Pleura in Gebhard Mathis (Edn) Chest Sonography, SBN 978-3-540-72427-8 Springer Berlin Heidelberg New York.
- Qureshi NR, Rahman NM, Gleeson FV. Thoracic ultrasound in the diagnosis of malignant pleural effusion. Thorax. 64, 139- 143 (2009).
- Pinto C, Betta PG, Fava C et al. Second Italian consensus conference on malignant pleural mesothelioma: state of the art and recommendations. Cancer. Treat. Rev. 39, 328- 39 (2013).
- Armato SG, Labby ZE, Coolen J et al. Imaging in pleural mesothelioma: a review of the 11th International Conference of the International Mesothelioma Interest Group. Lung. Cancer. 82, 190-196 (2013).
- Hooper C, Lee YC, Maskell N. BTS Pleural Guideline Group. Investigation of a unilateral pleural effusion in adults: British Thoracic Society Pleural Disease Guideline. Thorax. 2, ii4-ii17 (2010).
- Stahel RA, Weder W, Lievens Y. ESMO Guidelines Working Group Malignant pleural mesothelioma: ESMO Clinical Practice Guidelines for diagnosis, treatment and followup. Ann. Oncol. 21( 5), v126-v128 (2010).
- van Zandwijk N, Clarke C, Henderson D et al. Guidelines for the diagnosis and treatment of malignant pleural mesothelioma. J. Thorac. Dis. 5, E254-307 (2013).