Research Article - Imaging in Medicine (2013) Volume 5, Issue 1
Evaluation of intima-media thickness of carotid artery in patients with chronic graft-versus-host disease using ultrasound
Mohammad Nazari1, Afsaneh Morteza1, Masoud Irvani2, Ardeshir Ghavamzadeh2, Arman Al-e-Esmael1, Omid Khalilzade3 & Hadi Rokni-Yazdi*11Advanced Diagnostic & Interventional Radiology Research Center (ADIR), Imaging Medical Center, Imam Hospital, Tehran University of Medical Sciences, Tehran, Iran
2Hematology, Oncology & Stem Cell Transplantation Research Center, Shariati Hospital, Tehran University of Medical Sciences, Tehran, Iran
3Department of Radiology, Massachusetts General Hospital, Harvard Medical School, MA, USA
- Corresponding Author:
- Hadi Rokni-Yazdi
Advanced Diagnostic & Interventional Radiology Research Center (ADIR)
Imaging Medical Center, Imam Hospital
Tehran University of Medical Sciences, Tehran, Iran
Tel: +98 216 119 2670
Fax: +98 216 690 201
E-mail: hadirokni@yahoo.com
Abstract
Aim: We studied the use of ultrasound in the detection of intima-media thickness (IMT) as an early predictor of atherosclerosis in patients with and without chronic graft-versus-host disease (GVHD) after allogeneic bone marrow transplantation (BMT).
Methods: We performed a cross-sectional study of 30 BMT patients without history of chronic GVHD, 30 BMT patients with chronic GVHD and 30 healthy controls. IMT was estimated at 3 points on the right and left sides – 1: within 10 mm of the distal common carotid artery; 2: within 10 mm of the carotid bulb; and 3: within 10 mm of the proximal internal carotid artery.
Results: Patients with chronic GVHD had a significantly (p < 0.01) higher overall IMT and IMT in the right or left bulb of carotid compared with patients without chronic GVHD and controls. No differences were found in IMT at different points between controls and patients without GVHD.
Conclusion: This study demonstrated a higher IMT in patients with chronic GVHD after BMT and no significant difference in IMT of patients without GVHD compared with controls.
Keywords
allogeneic bone marrow transplantation ▪ carotid artery ▪ graft-versus-host disease ▪ intima-media thickness ▪ ultrasonography
Allogeneic bone marrow transplantation (BMT) is an effective and curative treatment for a wide range of malignant and non-malignant hematological disorders [1–3]. Studies following patients who underwent BMT for 2–3 decades have demonstrated considerable long-term morbidities and mortalities in these patients [4]. Graft-versus-host disease (GVHD) is a common complication that can occur after bone marrow or stem cell transplantation. In GVHD, newly transplanted cells attack the transplant recipient’s body [5,6]. Acute GVHD usually occurs within the first 3 months after transplantation, whereas chronic GVHD usually begins more than 3 months after transplantation, and can last a lifetime [5,6]. Chronic GVHD is the primary cause of transplant-related mortality after BMT and contributes to most of its non-malignant complications [7]. Although prophylactic treatments have been introduced, the incidence of chronic GVHD has remained constant [7,8].
Atherosclerosis and cardiovascular disorders are among the life-threatening complications affecting individuals with chronic GVHD after BMT [3]. Nowadays, atherosclerosis is defined as inflammation of the arterial wall, which ultimately leads to lipid storage in the endothelium and further damage [9]. Chronic GVHD is an inf lammatory state in which immune-mediated inf lammation causes endothelial damage [10–12]. It is reported that in BMT patients, cardiovascular events have a more dramatic course and can be observed in unexpectedly young patients [13–17]. Hence, detection of atherosclerosis after BMT is of great clinical importance. Ultrasound has demonstrated valuable promise in detection of IMT [18]; however, there is limited evidence in patients with chronic GVHD. The aim of this study was to evaluate, for the first time, the use of ultrasound in the detection of IMT in patients with and without chronic GVHD, as well as in healthy controls.
Methods
Participants
We performed a cross-sectional study of 30 patients undergoing consecutive BMT without history of chronic GVHD, which included 30 BMT patients with chronic GVHD and 30 healthy controls. The study was conducted from October 2009 to January 2011 in the Advanced Diagnostic and Interventional Radiology Research Center, affiliated with Tehran University of Medical Sciences. Patients with BMT were selected from the hematology clinic of Shariati Hospital affiliated with Tehran University of Medical Sciences. Chronic GVHD was diagnosed according to: classic chronic GVHD (without features or characteristics of acute GVHD) and an overlap syndrome in which diagnostic or distinctive features of chronic GVHD and acute GVHD appear together [5]. In our patients, allogeneic BMT was performed according to the protocol described by Slavin et al. [19]. Inclusion criteria were: allogeneic BMT duration of more than 365 days and being aged between 18 and 35 years old. The patients with and without GVHD were matched according to BMT duration. Exclusion criteria were: diabetes mellitus; hypertension; obesity (BMI ≥30 kg/m2); cigarette smoking; chronic renal failure; history of cerebrovascular accidents; dyslipidemia; and coronary heart disease in patients and controls. Healthy controls were selected from the patient’s partners or hospital staff. Controls were matched with patients according to age and sex. The study was approved by the local ethics review committee at the university. After explaining the study process in detail to the patients and controls, written informed consent was obtained from all participants.
Ultrasound
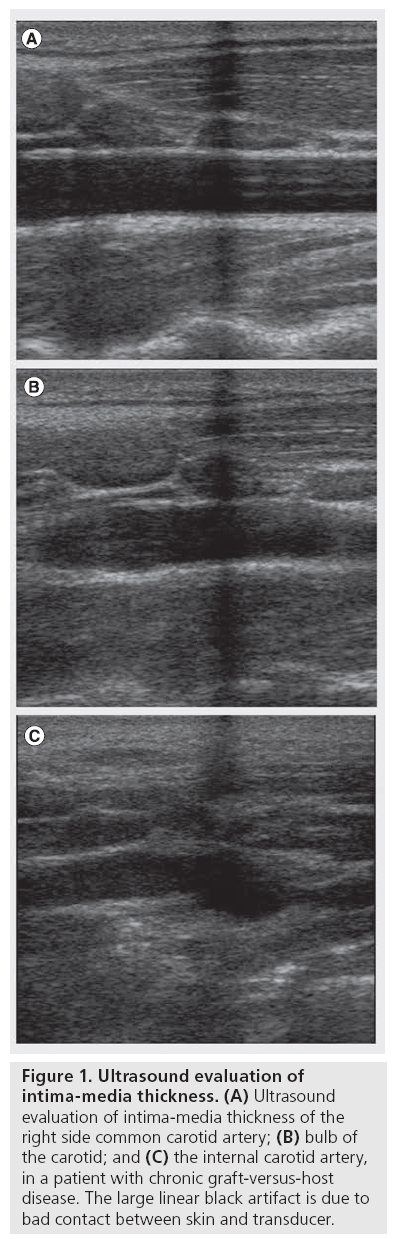
A single radiologist, blinded to both the patients and control group, performed the measurements using a B-mode ultrasound system equipped with a 5–10-MHz linear array transducer (SONOLINE G-40, Siemens, Erlangen, Germany). The most appropriate frequency was selected in each subject according to the best resolution. The transducer was applied to the neck skin surface of the participants, in the supine position, using an appropriate amount of ultrasound gel. Focus was adjusted on the far vessel wall of the carotid artery. In the longitudinal ultrasound planes of the carotid artery, three echogenic lines were detected in the near and far walls: at the interface between arterial lumen and intima (lumen–intima interface); at the interface between media and adventitia (media–adventitia interface); and at the interface between adventitia and periadventitia tissues (adventitia–periadventitia interface). The distance between the first and second echogenic lines was considered as the IMT [20]. IMT was measured at three points: within 10 mm of the distal common carotid arteries; within 10 mm of the carotid bulbs; and within 10 mm of proximal internal carotid arteries (Figure 1) [21]. All measurements were made manually with an electronic caliper from still images taken during real-time ultrasonography. The highest IMT at each point was measured. Measurements in each location were repeated on three different still images and were averaged. According to Yamasaki et al., the overall mean of IMT at different points was also calculated [22]. Plaques (defined either as focal mural protrusion into vessel lumen or as a definite echogenicity with posterior shadowing) were not included in IMT measurements [18].
Figure 1: Ultrasound evaluation of intima-media thickness. (A) Ultrasound evaluation of intima-media thickness of the right side common carotid artery; (B) bulb of the carotid; and (C) the internal carotid artery, in a patient with chronic graft-versus-host disease. The large linear black artifact is due to bad contact between skin and transducer.
Statistical analysis
Statistical analysis was performed using SPSS software (version 17.0, SPSS Inc., IL, USA). Continuous variables were expressed as the mean ± standard error of mean. Normally distributed continuous variables were compared between patients with and without GVHD and with healthy controls using one-way ANOVA, following post hoc tests (Tukey). General linear models were employed to compare the IMT mean between patients with and without GVHD after multiple adjustments for age, sex and duration of transplantation. A p-value of <0.05 was considered statistically significant.
Results
Characteristics of the study participants are presented in Table 1. Age and sex ratios were similar between healthy controls and patients with and without GVHD. Patients with and without GVHD did not significantly differ for different causes of BMT. No significant differences were found in IMT at different points between controls and patients without GVHD. Patients with chronic GVHD had a significantly (p < 0.01) higher overall IMT and IMT in the right or left bulb of the carotid compared with patients without chronic GVHD and controls (Table 1). The higher IMT mean in patients with chronic GVHD versus patients without GVHD remained significant after multiple adjustments for age, sex and duration of transplantation using general linear models (5.3 ± 0.14 vs 4.6 ± 0.14; p < 0.01).
Discussion
Our study clearly demonstrated that BMT patients with chronic GVHD had higher values of overall IMT and IMT in the bulb of the carotid compared with patients without chronic GVHD, or healthy controls. To date, we are unaware of any other study showing the applicability of ultrasound in the evaluation of IMT in patients with chronic GVHD.
Chronic GVHD is a relatively common complication after allogeneic BMT with an increasing incidence [23], secondary to the increasing use of BMT in older patients, transplants from unrelated and mismatched related donors, peripheral blood stem cell transplantations, and donor lymphocyte infusions, which carry a higher risk for the development of chronic GVHD [23–25].
Atherosclerosis is a chronic inflammatory disease that starts early in life [12]. Recent studies suggest that chronic GVHD could play a role in the development and progression of atherosclerosis and cardiovascular events [26–29]. What is the pathogenesis of atherosclerosis in chronic GVHD? Formerly it was believed that atherosclerosis is a consequence of the traditional cardiovascular risk factors after the hematopoietic bone marrow transplantation [30]. Recently it was demonstrated that the inflammation is the pathogenic mechanism linking atherosclerosis and chronic GVHD [12]. Microangiopathy, venoocclusive disease of the liver, diffuse alveolar hemorrhage, engraftment syndrome and capillary leak syndrome are the early endothelial events after BMT [12]. Recent studies have demonstrated that they are mainly caused by cellular toxicity, the procoagulant condition, loss of microvessels, perivascular infiltration of activated cytotoxic T lymphocytes and the inflammatory response after BMT [31]. All these events in patients with chronic GVHD suggests that factors related to the alloreactive immune reaction should be involved in the atherosclerotic process [3]. Vascular endothelial cells are an exposed target tissue, and endothelial damage in GVHD could contribute to atherosclerosis [10–12]. This process may be accelerated by increased levels of inflammatory markers in the circulation [32,33]. These events are enhanced by the traditional cardiovascular risk factors, such as diabetes, hypertriglyceridemia, low levels of high-density lipoprotein cholesterol, hypertension and obesity [30]. In this study, we aimed to evaluate the role of chronic GVHD as an independent factor in the development of atherosclerosis, therefore patients with known history of cardiovascular risk factors were excluded.
In our study, the higher IMT in patients with chronic GVHD versus those without GVHD indicates the association of chronic GVHD and atherosclerosis. Atherosclerosis is a side effect of BMT, which is mostly observed in young patients. The current study only includes patients from 18 to 35 years old so their long-term survival is very important. The noninvasive and widely available diagnostic tools for early diagnosis of atherosclerosis should be considered in these patients. Here we showed that ultrasonography could be used for early diagnosis of atherosclerosis in these patients. Consistent with our findings, increased IMT has been used as a marker of atherosclerosis in people without BMT [34,35], and is also is an early marker predicting cerebrovascular events [36]. We suggest that ultrasonoraphy could be considered as a screening tool in patients who underwent BMT, without chronic GVHD.
Future perspective
The principal limitation of the present study is that ultrasonography is an operator-dependent imaging tool. In our study a single radiologist, who was blinded to the patient and control groups, performed all the ultrasound examinations, and there is no evidence for the repeatability and reproducibility of these measurements. Computer-aided design, the use of computer systems to assist the creation and modification or optimization of the measurements, should be considered for future studies. Furthermore, we performed a cross-sectional study, and therefore the causal pathways underlying the observed relationships cannot be inferred. Future prospective studies may elucidate the role of ultrasound in the early prediction of cardiovascular disorders in patients with chronic GVHD after BMT. On the other hand, we took advantage of a relatively large sample size and close similarity between groups in most of the potentially confounding variables. In conclusion, our findings demonstrated the applicability of ultrasound in the detection of a higher IMT in patients with chronic GVHD after BMT. Our results may indicate the usefulness of ultrasound for detection of increased IMT and follow-up of these patients.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
Financial & competing interests disclosure
This study is supported financially by a grant from Tehran University of Medical Sciences. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conf lict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
References
- Brunner S, Huber BC, Fischer R et al. G-CSF treatment after myocardial infarction: impact on bone marrow-derived vs cardiac progenitor cells. Exp. Hematol. 36(6), 695–702 (2008).
- Tichelli A, Passweg J, Wojcik D et al. Late cardiovascular events after allogeneic hematopoietic stem cell transplantation: a retrospective multicenter study of the Late Effects Working Party of the European Group for Blood and Marrow Transplantation. Hematologica 93(8), 1203–1210 (2008).
- Tichelli A, Bucher C, Rovo A et al. Premature cardiovascular disease after allogeneic hematopoietic stem-cell transplantation. Blood 110(9), 3463–3471 (2007).
- Baker KS, Gurney JG, Ness KK et al. Late effects in survivors of chronic myeloid leukemia treated with hematopoietic cell transplantation: results from the Bone Marrow Transplant Survivor Study. Blood 104(6), 1898–1906 (2004).
- Filipovich AH, Weisdorf D, Pavletic S et al. NIH consensus development project on criteria for clinical trials in chronic graftversus- host disease: I. Diagnosis and staging working group report. Biol. Blood Marrow Transplant 11(12), 945–956 (2005).
- Fraser CJ, Bhatia S, Ness K et al. Impact of chronic graft-versus-host disease on the health status of hematopoietic cell transplantation survivors: a report from the Bone Marrow Transplant Survivor Study. Blood 108(8), 2867–2873 (2006).
- Vogelsang GB. How I treat chronic graft-versus-host disease. Blood 97(5), 1196–1201 (2001).
- Lee SJ, Klein JP, Barrett AJ et al. Severity of chronic graft-versus-host disease: association with treatment-related mortality and relapse. Blood 100(2), 406–414 (2002).
- Spirig R, Tsui J, Shaw S. The emerging role of TLR and innate immunity in cardiovascular disease. Cardiol. Res. Pract. 2012, 181394 (2012).
- Pober JS. Warner-Lambert/Parke-Davis award lecture. Cytokine-mediated activation of vascular endothelium. Physiology and pathology. Am. J. Pathol. 133(3), 426–433 (1988).
- Biedermann BC. Vascular endothelium and graft-versus-host disease. Best Pract. Res. Clin. Hematol. 21(2), 129–138 (2008).
- Tichelli A, Gratwohl A. Vascular endothelium as ‘novel’ target of graft-versus-host disease. Best Pract. Res. Clin. Hematol. 21(2), 139–148 (2008).
- Gatt ME, Liebster D, Leibowitz D, Matzner Y. Acute myocardial infarction after bone marrow transplantation: an unsuspected late complication. Ann. Hematol. 82(2), 136–138 (2003).
- Ghobrial IM, Bunch TJ, Caplice NM, Edwards WD, Miller DV, Litzow MR. Fatal coronary artery disease after unrelated donor bone marrow transplantation. Mayo. Clin. Proc. 79(3), 403–406 (2004).
- Gordon BG, Saving KL, McCallister JA et al. Cerebral infarction associated with protein C deficiency following allogeneic bone marrow transplantation. Bone Marrow Transplant. 8(4), 323–325 (1991).
- Uchida N, Taniguchi S, Harada N, Shibuya T. Myocardial ischemia following allogeneic bone marrow transplantation: possible implication of tacrolimus overdose. Blood 96(1), 370–372 (2000).
- van der Lelie J, Louwerse ES, Thomas LL, van Oers MH, von dem Borne AE. Acute ischemic cerebrovascular accident after autologous bone marrow transplantation. Eur. J. Hematol. 56(1–2), 95–97 (1996).
- Keser G, Aksu K, Tamsel S et al. Increased thickness of the carotid artery intima-media assessed by ultrasonography in Behcet’s disease. Clin. Exp. Rheumatol. 23(4 Suppl. 38), S71–S76 (2005).
- Slavin S, Nagler A, Naparstek E et al. Non-myeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and non-malignant hematologic diseases. Blood 91(3), 756–763 (1998).
- Pignoli P, Tremoli E, Poli A, Oreste P, Paoletti R. Intimal plus medial thickness of the arterial wall: a direct measurement with ultrasound imaging. Circulation 74(6), 1399–1406 (1986).
- Touboul PJ, Hennerici MG, Meairs S et al. Mannheim intima-media thickness consensus. Cerebrovasc. Dis. 18(4), 346–349 (2004).
- Yamasaki Y, Kodama M, Nishizawa H et al. Carotid intima-media thickness in Japanese type II diabetic subjects: predictors of progression and relationship with incident coronary heart disease. Diabetes Care 23(9), 1310–1315 (2000).
- Socie G, Salooja N, Cohen A et al. Non-malignant late effects after allogeneic stem cell transplantation. Blood 101(9), 3373–3385 (2003).
- Higman MA, Vogelsang GB. Chronic graft versus host disease. Br. J. Hematol. 125(4), 435–454 (2004).
- Cutler C, Giri S, Jeyapalan S, Paniagua D, Viswanathan A, Antin JH. Acute and chronic graft-versus-host disease after allogeneic peripheral-blood stem-cell and bone marrow transplantation: a meta-analysis. J. Clin. Oncol. 19(16), 3685–3691 (2001).
- Fassas A, Passweg JR, Anagnostopoulos A et al. Hematopoietic stem cell transplantation for multiple sclerosis. A retrospective multicenter study. J. Neurol. 249(8), 1088–1097 (2002).
- Baker KS, Ness KK, Steinberger J et al. Diabetes, hypertension, and cardiovascular events in survivors of hematopoietic cell transplantation: a report from the bone marrow transplantation survivor study. Blood 109(4), 1765–1772 (2007).
- Armenian SH, Bhatia S. Cardiovascular disease after hematopoietic cell transplantation – lessons learned. Hematologica 93(8), 1132–1136 (2008).
- Halter J, Kodera Y, Ispizua AU et al. Severe events in donors after allogeneic hematopoietic stem cell donation. Hematologica 94(1), 94–101 (2009).
- Gratwohl A, Brand R, Apperley J et al. Allogeneic hematopoietic stem cell transplantation for chronic myeloid leukemia in Europe 2006: transplant activity, longterm data and current results. An analysis by the Chronic Leukemia Working Party of the European Group for Blood and Marrow Transplantation (EBMT). Hematologica 91(4), 513–521 (2006).
- Biedermann BC, Sahner S, Gregor M et al. Endothelial injury mediated by cytotoxic T lymphocytes and loss of microvessels in chronic graft versus host disease. Lancet 359(9323), 2078–2083 (2002).
- Deeg HJ. Graft-versus-host disease: host and donor views. Semin. Hematol. 30(4 Suppl. 4), 110–117; discussion 118 (1993).
- Howell CD, Li J, Chen W. Role of intercellular adhesion molecule-1 and lymphocyte function-associated antigen-1 during nonsuppurative destructive cholangitis in a mouse graft-versus-host disease model. Hepatology 29(3), 766–776 (1999).
- Coll B, Feinstein SB. Carotid intima-media thickness measurements: techniques and clinical relevance. Curr. Atheroscler. Rep. 10(5), 444–450 (2008).
- De Groot E, Van Leuven SI, Duivenvoorden R et al. Measurement of carotid intima-media thickness to assess progression and regression of atherosclerosis. Nat. Clin. Pract. Cardiovasc. Med. 5(5), 280–288 (2008).
- Saba L, Mallarini G, Sanfilippo R, Montisci R, Suri JS. Intima media thickness variability (IMTV) and its association with cerebrovascular events: a novel marker of carotid atherosclerosis? Cardiovasc. Diagn. Ther. 2, 10–18 (2012).