Review Article - Interventional Cardiology (2010) Volume 2, Issue 2
Impact of percutaneous patent foramen ovale closure on migraine course
- Corresponding Author:
- Bernhard Meier
Cardiology, University Hospital
University of Bern, 3010 Bern, Switzerland
Tel: + 41 31 632 3077
Fax: +41 31 382 1069
E-mail: bernhard.meier@insel.ch
Abstract
Keywords
migraine, patent foramen ovale, percutaneous shunt closure, shunt, treatment
In 1942, a distinguished Havard professor of physiology published a paper entitled ‘voodoo death’ [1]. The paper reported anecdotal experiences of death from fright and was an early example of the ‘brain–heart connection’. The term ‘brain–heart connection’ includes the heart’s effects on the brain (e.g., cardiac sources of embolic stroke) and the brain’s effects on the heart (e.g., ECG abnormalities in patients with subarachnoid hemorrhage) [2]. The first thoughts of neurologists in response to preliminary reports suggesting an interaction of intracardiac shunts with migraine may have included attributes such as voodoo and unwittingly but correctly pointed to a brain–heart connection [3–5].
This article summarizes the current knowledge regarding migraine pathophysiology and the data regarding the impact of patent foramen ovale (PFO) closure on migraine. We continue to have a limited understanding of the mechanisms of how a PFO and migraine may be inter-related, and the concepts regarding migraine pathophysiology in the setting of a PFO that we discussed recently [6] have neither been proven nor refuted yet.
Migraine
Migraine is a recurrent, disabling neurological disorder affecting 8–13% of the adult population [7]. Migraine is three-times more common in women and its prevalence peaks at 40 years of age [8]. The main subcategories are migraine with and without aura [9].
Migraine without aura manifests in headache attacks that last 4–72 h, typically have a unilateral location, are of pulsating quality, moderate or severe intensity and are aggravated by physical activity. During the migraine headache, nausea or vomiting and photophobia or phonophobia are present. History, physical examination and neurologic examination do not suggest any underlying organic disease. Migraine with aura manifests in attacks of reversible focal neurologic symptoms that develop gradually over 5–20 min and last for less than 60 min. A headache with the features of migraine without aura begins during the aura or follows the aura within 60 min. Other terms for migraine with aura are classic migraine, ophthalmic, hemiparaesthetic, hemiplegic or aphasic migraine, migraine accompagnée and complicated migraine.
Two-thirds of migraine patients have exclusively migraine without aura, the remainder have migraine with aura or both types of migraine. A typical migraineur has one to two attacks per month, one in every ten migraineurs has weekly attacks, and two in ten have attacks lasting 2 or 3 days. Most patients with a migraine aura describe visual disturbances as the leading symptom. In 1944, Leão described a phenomenon termed cortical spreading depression, now believed to be the pathophysiology of an aura attack [10]. He observed in rodents after noxious stimulation of the brain a spreading decrease in cortical electrical activity. This was due to a cortical propagating wave of neuronal depolarization followed by prolonged inhibition of neuronal activity [11]. The propagating neuronal inhibition produces an aura phenomenon and is accompanied by a decrease in cortical blood flow [12]. This reduction in cerebral perfusion very rarely causes manifest ischemia. Hence, contrary to the once popular vascular theory, the aura phenomenon is not caused by cerebral vasoconstriction and ischemia due to a dysfunctional cerebral vasoregulation, but indeed by dysfunctional neurons provoking a decrease in cortical blood flow [8]. The cortical spreading depression wave finally activates neurons in the trigeminal ganglion and nucleus and this trigeminal activation contributes to the migraine headache [13]. Headache results from activation of meningeal and blood vessel nociceptors combined with a change in central pain modulation. In patients with migraine without aura, the phenomenon of cortical spreading depression may occur in silent areas of the cortex or the cerebellum [14].
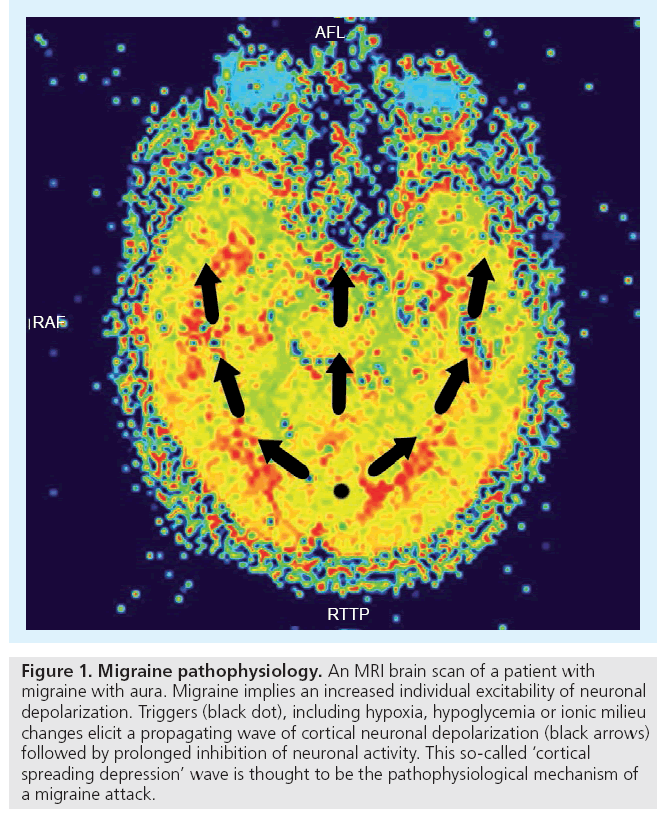
Not all of us suffer from migraine, since not all of us have an increased neuronal excitability to react on different triggers by a cortical spreading depression wave. In other words, migraine implies an individual susceptibility (Figure 1).
Figure 1: Migraine pathophysiology. An MRI brain scan of a patient with migraine with aura. Migraine implies an increased individual excitability of neuronal depolarization. Triggers (black dot), including hypoxia, hypoglycemia or ionic milieu changes elicit a propagating wave of cortical neuronal depolarization (black arrows) followed by prolonged inhibition of neuronal activity. This so-called ‘cortical spreading depression’ wave is thought to be the pathophysiological mechanism of a migraine attack.
In daily life, migraine attacks can be triggered by a multitude of events such as emotional stress, lack of sleep or oversleeping, dietary constituents such as chocolate, wine or caffeine, physical stimuli such as bright or flickering light, alterations in barometric pressure, medications or hormonal changes (e.g., menstruation). In the laboratory setting, cortical spreading depression can be initiated by hypoxia, hypoglycemia, mechanical trauma or changes in the extracellular ionic milieu [15]. Cerebral ischemia is a trigger for migraine attacks [16] and ischemia-induced symptomatic migraine attacks are probably much more common than migraine-induced ischemic insults [17].
Patent foramen ovale
Galen already knew of the foramen ovale and its normal postnatal closure, but it was Leonardo Botallo, an Italian surgeon after whom the aorto–pulomonary duct was named, who described in the 16th Century the persistence of a foramen ovale after birth, without understanding its function in the fetus [18]. In a contemporary study with 965 autopsy specimens from patients without known cardiac disease, the foramen ovale was patent in 27% of cases [19]. In this often-cited necropsy study, the incidence of a PFO decreased with the patient’s age, suggesting that spontaneous PFO closure after birth may be possible or that selective mortality may offer an alternative explanation. The PFO incidence was 34% during the first three decades of life, 25% during the 4th through 8th decades and up to 20% during the 9th and 10th decades. In a recent letter from the Cleveland Clinic, an incidental PFO was found on an intraoperative transesophageal echocardiogram in 17% of 13,261 patients undergoing cardiac surgery, and this incidence was constant with age, suggesting that there may be no decrease in PFO incidence with age [20].
Discrepancies in the PFO prevalence among different studies depend on the methods used to diagnose this intracardiac shunt. In vivo, transthoracic or transesophageal echocardiography and transcranial Doppler studies, all with contrast bubbles, are used to detect this intermittent right-to-left shunt. In a comparative study in patients with a previous ischemic stroke, transesophageal contrast echocardiography was more sensitive than a transcranial Doppler examination in detecting a PFO, especially in cases of minimal right-to-left shunts. Transthoracic contrast echocardiography was the least sensitive test [21]. Diagnosing a PFO depends not only on the imaging method, but also on the ability of the patient to perform a Valsalva maneuver. The temporary raise in venous return and the concomitant increase in right atrial pressure after release of the Valsalva strain phase are decisive for the opening of the flap valve in most patients with a PFO. Sedation, often necessary to perform a transesophageal echocardiogram, can compromise an effective Valsalva maneuver and may be one of the reasons for a false negative exam. Cubital administration of contrast bubbles is another reason for missing some PFOs [22]. Due to the cardiac anatomy, especially the eustachian valve, inferior and not superior vena cava blood is preferentially directed to the fossa ovalis. In some patients, a real washout effect impedes the contrast bubbles arriving from the superior vena cava to cross the foramen ovale, even when it is widely open. Therefore, it is not surprising that the PFO prevalence is lower with imaging methods compared with necropsy studies.
Heart–brain connection
Transient global amnesia (TGA) is a clinical syndrome of abrupt reversible anterograde amnesia, accompanied by repetitive questioning. In most patients, TGA is precipitated by psychological factors (e.g., emotional stress and pain) or physical activities including a Valsalva maneuver. Intermittent cerebral ischemia or a cortical spreading depression wave have bee n proposed as the mechanism responsible for the transient amnesia [23]. In 1996, Klotzsch et al., using contrast transcranial Doppler sonography, observed a right-to-left shunt in 55% of the patients with TGA, compared with 27% of a control group [24]. In half of the TGA patients with a right-to-left shunt, a precipitating activity, such as lifting heavy weights or sexual intercourse occurred immediately before the amnesia.
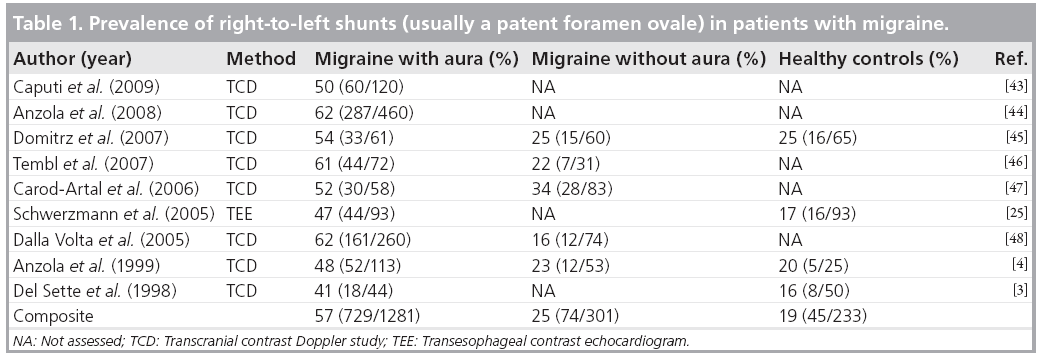
Based on clinical and pathophysiological similarities between TGA and migraine with aura, Anzola et al. decided to investigate the frequency of right-to-left shunts in a consecutive cohort of 113 migraineurs referred to their headache outpatient clinic [4]. The prevalence of a right-toleft shunt in their transcranial contrast Doppler studies was 48% in patients with migraine with aura, 23% in patients with migraine without aura, and 20% in age-matched control subjects. This difference between patients with and without migraine was significant (odds ratio [OR]: 3.13; 95% CI: 1.41–7.04) as was the difference between patients with migraine with aura and controls (OR 3.66; 95% CI: 1.21–13.25). Using transesophageal echocardiography instead of transcranial Doppler studies, we were able to document a PFO in half of all patients with migraine with aura, and also confirmed a PFO and not, for example, intrapulmonary fistulae or septal defects, in the overwhelming majority of patients with migraine and a shunt [25]. Table 1 provides an overview on the most recent studies assessing the prevalence of a PFO in migraine patients. Despite different imaging technologies, these studies consistently showed a PFO prevalence of 50–60% in over 1200 patients with migraine with aura. The prevalence of a PFO in patients with migraine without aura was similar to the prevalence of a PFO in controls.
There is also a correlation between the probability of migraine with aura and the size of the PFO. By directly assessing the PFO size at transesophageal echocardiography, we demonstrated that migraineurs with a PFO have a larger shunt size than controls with a PFO [25]. Anzola et al., analyzing shunt size with transcranial Doppler in 420 individuals with migraine, a history of stroke, or nonmigraineurs controls, found twice as large right-to-left shunts in migraineurs compared with nonmigraineurs. The largest shunts were observed in patients with migraine and a history of stroke. Patients with migraine and no stroke still had larger shunts than patients with a history of stroke but no migraine [26].
The brain–heart connection in migraine with aura is further strengthened by the observation that migraine with aura is also more prevalent in patients with a right-to-left shunt, independent of shunt anatomy. In divers with a history of decompression illness and an intracardiac shunt, Wilmshurst et al. reported migraine with aura symptoms unconnected with diving in nearly half of the subjects [27]. In addition, more than half of all patients with intrapulmonary shunts due to hereditary hemorrhagic teleangiectasia report typical migraine symptoms [28], and similarly, stroke patients with a PFO have migraine headaches more often in daily life (27% of 267 patients) than stroke patients without a PFO (14% of 314 patients) [29]. Migraine headaches are particularly frequent in the setting of a PFO in conjunction with an atrial septal aneurysm and, therefore, a large intracardiac shunt [30]. Of course, these data must be interpreted with caution because the typical migraine with aura patient is not a diver or a stroke patient, nor carrier of the gene responsible for hereditary hemorrhagic teleangiectasia. One can argue that these selected patients are not representative for a typical migraineur, and the increased migraine prevalence in these patients may not be related to the presence of a right-to-left shunt but to the patients’ underlying habits or disease.
So far, only one population-based study has been conducted to assess the relation of a PFO and migraine [31]. Out of 1101 stroke-free individuals from Northern Manhattan undergoing transthoracic echocardiography, 15% of patients had a PFO and 16% of patients declared to have migraine. The mean age of the cohort was 69 ± 10 years and 58% were women. The prevalence of a PFO was similar in patients with and without migraine. This study has some major flaws; the imaging method to detect a PFO has a low diagnostic accuracy and it is very likely that this study could not gather data from typical migraine patients. Migraine prevalence peaks at 40 years of age and, considering the age-specific migraine prevalence, patients aged in their 60s and 70s represent less than a fifth of all migraineurs and are not the typical migraine patient. This selection bias may explain the fact that 140 of 178 patients declared to have migraine with aura, whereas in nationwide epidemiological studies the majority of patients with migraine have migraine without aura.
In an attempt to quantify the current evidence regarding PFO and migraine, Schwedt and colleagues performed a quantitative systematic review of the literature and used metaanalysis statistics to combine the results of different studies [32]. Selecting 11 studies based on predefined criteria, the OR of migraine in a group of patients with PFO compared with the odds of migraine in a group of patients without a PFO was 5.13 (95% CI: 4.67–5.59). The OR of PFO in a group of patients with migraine compared with the OR of PFO in a group of patients without migraine was 2.54 (95% CI: 2.01–3.08). To date, this is probably the most precise estimate of the extent of the association between PFO and migraine.
How may PFO closure affect migraine?
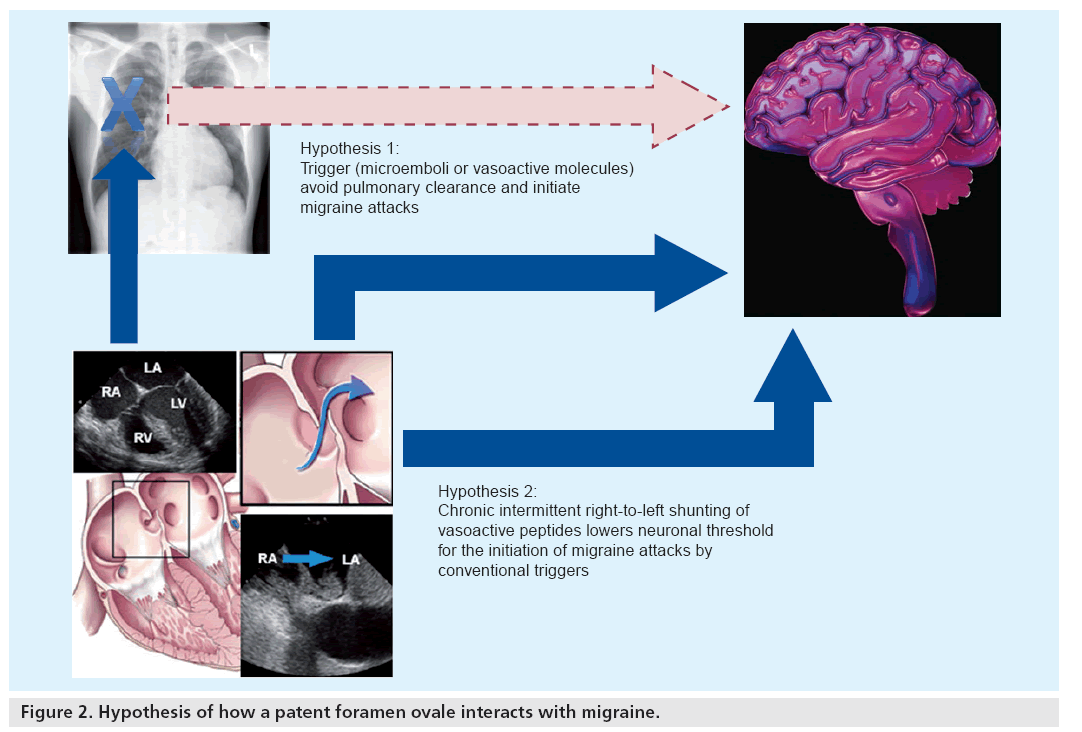
If one accepts that a PFO plays a pivotal role in the course of a migraine attack, the following scenarios are possible: given that the concept of migraine is based on an increased neuronal excitability and the presence of migraine triggers, a PFO can either allow solid or metabolic migraine triggers to avoid pulmonary clearance and then hit sensitive regions of the cortex, and thereby initiate a migraine attack. Alternatively, a PFO can act as a sensitizer of neurons to respond to any trigger with the initiation of a cortical spreading depression wave. Chronic shunting may repetitively expose occipital neurons to microemboli or vasoactive peptides, thereby lowering their ‘neuronal threshold’ (Figure 2). Of all these theories, the possibility of microemboli as triggers of migraine attacks is probably the most likely as it is supported by the majority of current literature. Box 1 summarizes the current arguments favoring the embolic hypothesis.
Metabolic right-to-left shunting has been proposed as an alternative trigger of migraine attacks. Serotonin, usually metabolized by the pulmonary monoamine oxidase, can partially escape pulmonary clearance in the presence of a right-to-left shunt. The exact role of serotonin in migraine is not clear, but drugs designed to specifically antagonize a subgroup of serotonin receptors (i.e., triptans) can abort migraine attacks. Serotonin may either activate platelets to aggregate more easily and hence lead to cerebral infarction, or may by itself initiate cortical spreading depression by binding to specific receptors.
All theories imply that it is not the PFO per se, but rather the right-to-left shunt that accounts for migraine. The fact that closure of pulmonary fistulae is also associated with migraine improvement underscores the concept that not the anatomy, but the functional impact of right-to-left shunts is associated with migraine [33].
Impact of shunt closure on migraine course
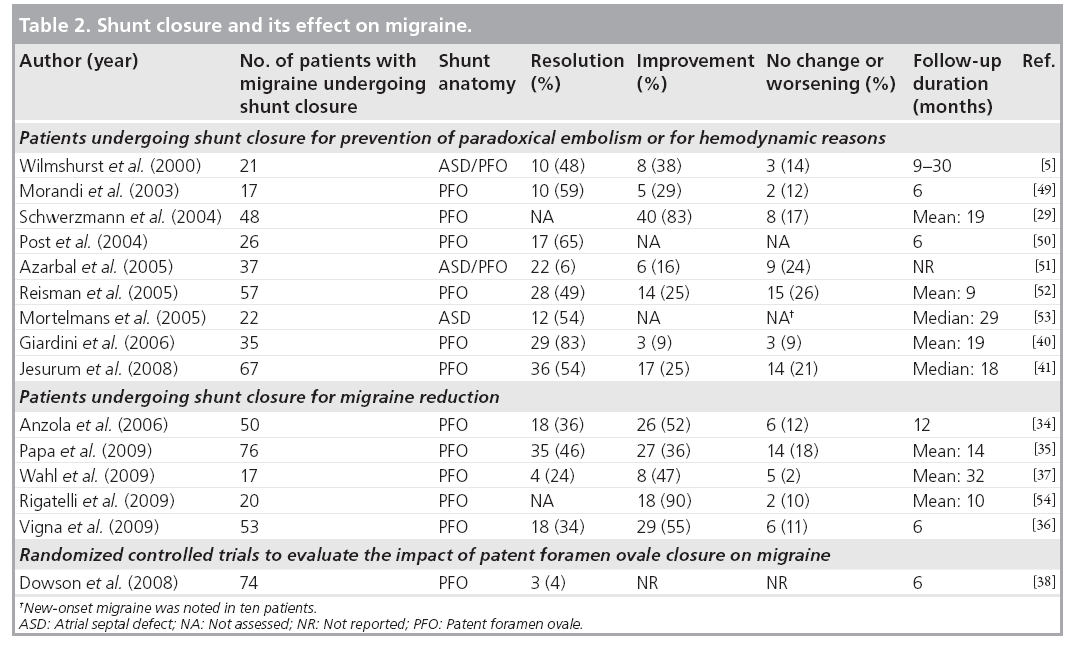
Since the first report of an improvement in migraine after intracardiac shunt closure in 2000 [5], more than a dozen additional reports have been published, all in support of a PFO–migraine connection. The first reports of this series had patients undergoing shunt closure for other reasons than migraine prevention and showed an unexpected reduction in migraine frequency during follow-up. These unexpected findings were followed by nonrandomized studies addressing the impact of PFO closure on headache severity in selected migraine patients undergoing shunt closure to improve their migraine. The third, and unfortunately smallest, category of publications consists of one randomized, blinded (sham procedure) and controlled trial investigating the impact of PFO closure on migraine intensity. Table 2 provides an overview of all these reports.
Wilmshurst and colleagues were the first to notice an impressive reduction of migraine frequency in patients undergoing percutaneous atrial shunt closure for prevention of paradoxical embolism or for hemodynamic reasons [5]. The majority of the 32 patients were divers with a history of decompression illness events. A total of 21 of the patients had migraine (16 with aura) before shunt closure. After the intervention, migraine did not reoccur in seven patients, with other patients experiencing on average 6–12 migraine attacks per year. Only three of the 21 migraineurs had no improvement in headache severity and frequency during a follow- up of up to 30 months.
Retrospectively analyzing our own PFO closure patients, we were able to assess headache characteristics in 215 of them before and after PFO closure [29]. Our study cohort was older than Wilmshurst’s cohort (mean age 50 vs 35 years, respectively) and the majority of our patients were suspected to have had paradoxical cerebral embolism. We classified patients with concomitant headaches into three categories, specifically, migraine with (n = 37) and without aura (n = 11) and tension-type headache (n = 23). PFO closure significantly reduced migraine attack frequency by more than 50%. Migraine patients with and without aura had a significant improvement in headache frequency, but not patients with tension-type headaches. The mean follow-up duration was 1.7 years and exceeded the 6 months postimplantation period with antiplatelet therapy in all patients. By routine repetition of transesophageal echocardiography 6 months after the intervention, we were able to show that three out of seven patients with no migraine improvement after shunt closure indeed had a residual shunt.
In 2006, Anzola et al. published a case–control study on the effects of shunt closure in migraineurs [34]. In a nonrandomized fashion, they selected 23 patients with migraine, a PFO and a stroke confirmed by neuroimaging and 27 patients with migraine, a PFO and no stroke, to undergo PFO closure. In addition, 27 patients with migraine and a PFO and no intervention were used as a control group. After 1 year of follow- up, migraine severity assessed with specific migraine scores decreased significantly in both intervention groups whereas no improvement was found in the control group. The reduction in migraine severity was independent of the type of migraine (with or without aura), the patient’s age and cerebrovascular risk factors. At the end of the study, 18 of 51 patients with migraine undergoing PFO closure were reported to be cured from migraine. Another Italian group performed PFO closure in 76 highly symptomatic migraine patients with a large PFO and clinical or subclinical ischemic brain lesions detected by MRI [35]. Beside migraine headaches, 16 patients had a history of stroke, and 32 patients had a history of recurrent transient ischemic attacks. After a mean follow-up of 14 months, no further cerebrovascular episode had occurred. Migraine was completely abolished in 35 patients (46%), improved in 27 (36%) and unchanged in 14 (18%). These astonishing results were further confirmed by another study in migraine patients with PFO and now exclusively asymptomatic ischemic brain lesions [36]. A total of 53 patients consented to undergo PFO closure and were prospectively followed for 6 months. Another group of 29 patients preferred to continue with medical therapy and served as a control group. At the end of the study period, migraine was completely resolved in 34% of the closure group and 7% of the controls (p < 0.001). Disabling migraine headaches disappeared in 53% of the closure group patients and 7% of the controls (p < 0.001). Furthermore, our own experience suggests that PFO closure impressively reduces the burden of migraine headaches [37]. The effects of PFO closure on migraine by far exceed the efficacy of medical therapy in migraine prevention.
Uncleared mist
Based on these encouraging nonrandomized observations, NMT Medical Inc. (MA, USA) funded a prospective, randomized, double-blind and sham-controlled trial to study the effects of PFO closure with their STARFlex® device on migraine with aura (Migraine Intervention with STARflex Technology [MIST] trial) [38]. The study was carried out in the UK in 2005. More than 440 patients with migraine with aura were screened for the presence of a right-to-left shunt with transthoracic echocardiography. Only migraineurs with refractory migraine symptoms for at least 1 year (defined as nonresponding to two types of preventive medication), 5–23 migraine days per month and no history of cerebrovascular events were included. In the end, 147 patients with a moderate or large PFO underwent 1:1 randomization to a sham procedure or PFO closure. Sham procedure and device implantation were performed under general anesthesia. The sham procedure consisted of a small groin incision. All participants had acetylsalicyl acid and clopidogrel for 3 months. After a 6‑month follow-up period, the migraine burden was analyzed by a blinded headache specialist and transthoracic echocardiography was repeated to assess the residual shunt rate. The primary study end point was defined as complete cessation of migraine. The secondary end point was a more than 50% reduction of headache days, the conventional end point in medical migraine prevention trials. The final publication of the results in spring 2008 was disappointing from a cardiologist’s perspective and sparked a controversy over the conclusions and the conduct of this trial.
At 6 months after the intervention, only three patients in the PFO-closure group were free of migraine. This number was equal to the number of patients in the sham procedure group. With respect to the secondary end point, things were less clear. In the first announcement of the results presented at the ACC 2006 meeting, a 50% reduction of headache days was achieved by 42% of patients in the closure arm and by 23% in the control arm (p < 0.04) [39]. In the final publication, the reduction in migraine days was no longer significant. The authors explained that two patients in the closure arm were responsible for 20% of all headache days during the analysis period and only with exclusion of these two patients would there be a significant reduction in migraine days between the two groups. In addition, it was disturbing that some of the original investigators refused to sign off the manuscript before it was submitted for publication and claimed that the rate of residual shunts in the closure group was far higher (>30%) than the reported rate of 6%.
Why did the MIST trial not reproduce the expected success? Three aspects might have been instrumental. First, patients selected for the MIST trial were a fundamentally different patient population than the patients analyzed in the retrospective reports. None of the nonrandomized reports was focussed on patients with refractory migraine and most of the patients in the previous reports had one or two migraine attacks per month, and not four to five as in the MIST trial. Second, the residual shunt rate may have negatively affected the outcome. By using transthoracic echocardiography with its limited sensitivity compared with transesophageal echocardiography to assess small interatrial shunts after 6 months, the authors may have underestimated the residual shunt prevalence. In some of the nonrandomized reports, including our own experience, a residual shunt was associated with persistence or reduced improvement of migraine [5,29,36,40]. However, there is also at least one report indicating the migraine relief may occur despite a residual right-to-left shunt [41]. Third, the short follow-up period after the intervention/sham procedure (3 months healing phase, 3 months analysis phase) may be another reason why the effects of device closure on migraine course could have been underestimated. Endothelialization of the PFO closure device may not be complete 3 months after implantation, there may still be a significant residual shunt rate, and withdrawing antiplatelet therapy after merely 3 months may favor local microembolic events on nonendothelialized parts of the closure device, and thereby potentially promote migraine headaches.
For the time being, we have to wait for the results of other ongoing trials in order to refute or accept the conclusions of the MIST trials. At present, the European Percutaneous Closure of PFO In Migraine with Aura (PRIMA) trial – a randomized prospective study, and the Northern American Prospective Randomized Investigation to Evaluate Incidence of Headache Reduction in Subjects with Migraine and PFO Using the Amplatzer® PFO Occluder Compared to Medical Management (PREMIUM) trial are enrolling patients, both using the Amplatzer PFO occluder as the closure device. These trials continue to include only migraine patients refractory to preventive therapy, but they need to have more headache-free days per month (usually >14 days) than in the MIST I trial and the follow-up period has been extended to 1 year. The PRIMA trial will not have a sham procedure arm. The feasibility of blinding the patient is questionable if echocardiography is part of the follow-up exams. Two other trials have prematurely stopped recruiting patients because of financial aspects. One of them was the MIST II trial, another NMT-funded study with their new BioStar® device. After having screened more than 1400 migraine patients, only ‘a handful’ patients met the stringent requirements for randomization. The other trial (Effect of Septal Closure of Atrial PFO on Events of Migraine with Premere™ [ESCAPE]) was sponsored by the company St Jude Medical (MN, USA) and used the Premere closure device. The initial plan was to include close to 500 patients, but after 2 years of screening it became clear that this number could never be achieved within a reasonable time.
Future perspective
It is questionable if PFO closure trials in migraine patients with frequent and refractory headaches despite advanced medical therapy will ever show an impressive benefit of shunt closure in the near future. This is probably one of the lessons we have learned from the MIST trial. As far as we can deduce from all the nonrandomized reports regarding PFO closure and migraine, the ‘conventional’ migraine patients with one to two migraine attacks per month, not necessarily refractory to medical treatment, are the ones that have a chance to get relieved from migraine attacks by shunt closure. Unfortunately, there is no randomized trial in such patients on the horizon. Neurologists have lost interest in such trials and the regulatory agencies are not keen in running any kind of risks. Even if PFO closure is a very easy procedure (Box 2), its invasive nature and the potential (albeit rare) complications are still used as an argument against its widespread use, especially in young and otherwise healthy patients. It is worth noting that this kind of arguing disregards the side effects of drug therapy for migraine prevention. The next best thing to a PFO closure trial in typical, uncomplicated migraine patients would be a randomized trial in migraineurs with a PFO and subclinical brain lesions detected by MRI, independent of their migraine frequency. Approximately every tenth patient with migraine with aura may have brain lesions [42]. Unfortunately, the screening costs of such a study will be exorbitantly high.
To date, the evidence linking PFO closure with migraine reduction is limited, but intriguing. The coming years will show if the PFO–migraine connection is only voodoo or if there is really a harmful brain–heart connection that can be cutoff by closing the PFO. We hope that 10 years from now, screening for a PFO will be part of the routine diagnostic work-up of patients with migraine, especially patients with migraine with aura, and certain patients with migraine and a PFO will be offered the option to undergo percutaneous shunt closure. It is worth keeping in mind that an uneventfully closed PFO in the realm of migraine incidentally excludes subsequent paradoxical strokes or myocardial infarction, and these are not only a trifle collateral benefit.
Financial & competing interests disclosure
Bernard Meier is the recipient of research grants and speaker fees from AGA Medical, Plymouth, MN, USA. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Executive summary
Pathophysiology of migraine
▪ Migraine implies an increased individual susceptibility to react on certain triggers with characteristic headaches.
▪ Cerebral ischemia is a trigger for migraine attacks. Ischemia-induced symptomatic migraine attacks are more common than migraine-induced ischemic insults.
▪ Any right-to-left shunt (including a patent foramen ovale [PFO]) can probably either allow migraine triggers to bypass pulmonary clearance and initiate a migraine attack or increase the individual susceptibility for migraine.
Heart–brain connection
▪ Migraine is more common in patients with a PFO; and a PFO is more common in patients with migraine.
▪ This epidemiological association is much stronger for migraine with aura than for migraine without aura.
▪ PFO size gradually increases from controls to patients with a history of stroke to migraineurs; patients with migraine and a history of stroke have the largest shunts.
Impact of shunt closure on migraine course
▪ Several nonrandomized studies demonstrated a marked reduction of migraine (with and without aura) after percutaneous PFO closure; this benefit was observed in divers, patients with previous paradoxical embolism and migraine patients (with and without ischemic brain lesions).
▪ To date, the only randomized study with blinded patients and a sham procedure did not find any improvement in migraine; there is controversy over the conduct and conclusions of this trial.
▪ The PRIMA trial and PREMIUM trial in Europe and North America, respectively, are currently enrolling patients with a PFO and migraine with aura refractory to medical treatment.▪ Future trials should include patients with milder forms of migraine, as did the nonrandomized studies.
References
Papers of special note have been highlighted as:
▪ of interest
- Cannon Wb: ‘Voodoo’ Death. Am Anthropologist 44, 169–178 (1942).
- Samuels Ma: The brain–heart connection. Circulation 116(1), 77–84 (2007).
- Del Sette M, Angeli S, Leandri M et al.: Migraine with aura and right-to-left shunt on transcranial doppler: a case–control study. Cerebrovasc. Dis. 8(6), 327–330 (1998).
- Anzola Gp, Magoni M, Guindani M, Rozzini L, Dalla Volta G: Potential source of cerebral embolism in migraine with aura: a transcranial doppler study. Neurology 52(8), 1622–1625 (1999).
- Wilmshurst Pt, Nightingale S, Walsh Kp, Morrison Wl: Effect on migraine of closure of cardiac right-to-left shunts to prevent recurrence of decompression illness or stroke or for haemodynamic reasons. Lancet 356(9242), 1648–1651 (2000).
- Schwerzmann M, Nedeltchev K, Meier B: Patent foramen ovale closure: a new therapy for migraine. Catheter Cardiovasc. Interv. 69(2), 277–284 (2007).
- Henry P, Auray Jp, Gaudin Af et al.: Prevalence and clinical characteristics of migraine in france. Neurology 59(2), 232–237 (2002).
- Silberstein Sd: Migraine. Lancet 363(9406), 381–391 (2004).
- Headache Classification Subcommittee of the International Headache Society: The International Classification of Headache Disorders: 2nd Edition. Cephalalgia. 24(Suppl. 1), 9–160 (2004).
- Leão Aap : Spreading depression of activity in the cerebral cortex. J. Neurophysiol. 7, 359–390 (1944).
- Lauritzen M: Pathophysiology of the migraine aura. The spreading depression theory. Brain 117(Pt 1), 199–210 (1994).
- Cutrer Fm, Sorensen Ag, Weisskoff Rm et al.: Perfusion-weighted imaging defects during spontaneous migrainous aura. Ann. Neurol. 43(1), 25–31 (1998).
- Bolay H, Reuter U, Dunn Ak, Huang Z, Boas Da, Moskowitz Ma: Intrinsic brain activity triggers trigeminal meningeal afferents in a migraine model. Nat. Med. 8(2), 136–142 (2002).
- Silberstein Sd: Recent developments in migraine. Lancet 372(9647), 1369–1371 (2008).
- Gorji A: Spreading depression: a review of the clinical relevance. Brain Res. Rev. 38(1–2), 33–60 (2001).
- Olesen J, Friberg L, Olsen TS et al.: Ischaemia-induced (symptomatic) migraine attacks may be more frequent than migraineinduced ischaemic insults. Brain 116(Pt 1), 187–202 (1993).
- Bousser Mg, Welch Km: Relation between migraine and stroke. Lancet Neurol. 4(9), 533–542 (2005).
- Fransson Sg: The botallo mystery. Clin. Cardiol. 22(6), 434–436 (1999).
- Hagen Pt, Scholz Dg, Edwards Wd: Incidence and size of patent foramen ovale during the first 10 decades of life: an autopsy study of 965 normal hearts. Mayo Clin. Proc. 59(1), 17–20 (1984).
- Hart Sa, Krasuski Ra: Incidence of asymptomatic patent foramen ovale according to age. Ann. Intern. Med. 150(6), 431–432 (2009).
- Di Tullio M, Sacco Rl, Venketasubramanian N, Sherman D, Mohr Jp, Homma S: Comparison of diagnostic techniques for the detection of a patent foramen ovale in stroke patients. Stroke 24(7), 1020–1024 (1993).
- Gin Kg, Huckell Vf, Pollick C: Femoral vein delivery of contrast medium enhances transthoracic echocardiographic detection of patent foramen ovale. J. Am. Coll. Cardiol. 22(7), 1994–2000 (1993).
- Quinette P, Guillery-Girard B, Dayan J et al.: What does transient global amnesia really mean? Review of the literature and thorough study of 142 cases. Brain 129(Pt 7), 1640–1658 (2006).
- Klotzsch C, Sliwka U, Berlit P, Noth J: An increased frequency of patent foramen ovale in patients with transient global amnesia. Analysis of 53 consecutive patients. Arch. Neurol. 53(6), 504–508 (1996).
- Schwerzmann M, Nedeltchev K, Lagger F et al.: Prevalence and size of directly detected patent foramen ovale in migraine with aura. Neurology 65(9), 1415–1418 (2005).
- Anzola Gp, Morandi E, Casilli F, Onorato E: Different degrees of right-to-left shunting predict migraine and stroke: data from 420 patients. Neurology 66(5), 765–767 (2006).
- Wilmshurst P, Nightingale S: Relationship between migraine and cardiac and pulmonary right-to-left shunts. Clin. Sci. (Lond.) 100(2), 215–220 (2001).
- Moussouttas M, Fayad P, Rosenblatt M et al.: Pulmonary arteriovenous malformations: cerebral ischemia and neurologic manifestations. Neurology 55(7), 959–964 (2000).
- Schwerzmann M, Wiher S, Nedeltchev K et al.: Percutaneous closure of patent foramen ovale reduces the frequency of migraine attacks. Neurology 62(8), 1399–1401 (2004).
- Lamy C, Giannesini C, Zuber M et al.: Clinical and imaging findings in cryptogenic stroke patients with and without patent foramen ovale: the pfo -asa study. Atrial septal aneurysm. Stroke 33(3), 706–711 (2002).
- Rundek T, Elkind Ms, Di Tullio Mr et al.: Patent foramen ovale and migraine: a cross-sectional study from the northern manhattan study (nomas). Circulation 118(14), 1419–1424 (2008).
- Schwedt Tj, Demaerschalk Bm, Dodick Dw: Patent foramen ovale and migraine: a quantitative systematic review. Cephalalgia 28(5), 531–540 (2008).
- Post Mc, Thijs V, Schonewille Wj et al.: Embolization of pulmonary arteriovenous malformations and decrease in prevalence of migraine. Neurology 66(2), 202–205 (2006).
- Anzola Gp, Frisoni Gb, Morandi E, Casilli F, Onorato E: Shunt-associated migraine responds favorably to atrial septal repair: a case–control study. Stroke 37(2), 430–434 (2006).
- Papa M, Gaspardone A, Fracasso G et al.: Usefulness of transcatheter patent foramen ovale closure in migraineurs with moderate to large right-to-left shunt and instrumental evidence of cerebrovascular damage. Am. J. Cardiol. 104(3), 434–439 (2009).
- Vigna C, Marchese N, Inchingolo V et al.: Improvement of migraine after patent foramen ovale percutaneous closure in patients with subclinical brain lesions: a case–control study. JACC Cardiovasc. Interv. 2(2), 107–113 (2009).
- Wahl A, Praz F, Findling O et al.: Percutaneous closure of patent foramen ovale for migraine headaches refractory to medical treatment. Catheter Cardiovasc. Interv. 74(1), 124–129 (2009).
- Dowson A, Mullen Mj, Peatfield R et al.: Migraine intervention with STARflex® technology (MIST) trial: a prospective, multicenter, double-blind, sham-controlled trial to evaluate the effectiveness of patent foramen ovale closure with STARflex septal repair implant to resolve refractory migraine headache. Circulation 117(11), 1397–1404 (2008).
- Tobis J: Management of patients with refractory migraine and PFO: is MIST I relevant? Catheter Cardiovasc Interv 72(1), 60–64 (2008).
- Giardini A, Donti A, Formigari R et al.: Transcatheter patent foramen ovale closure mitigates aura migraine headaches abolishing spontaneous right-toleft shunting. Am. Heart J. 151(4), 922 e921–925 (2006).
- Jesurum Jt, Fuller Cj, Kim Cj et al.: Frequency of migraine headache relief following patent foramen ovale ‘closure’ despite residual right-to-left shunt. Am. J. Cardiol. 102(7), 916–920 (2008).
- Kruit Mc, Van Buchem Ma, Hofman PA et al.: Migraine as a risk factor for subclinical brain lesions. JAMA 291(4), 427–434 (2004).
- Caputi L, D’amico D, Usai S, Grazzi L, Parati Ea, Bussone G: Prevalence and characteristics of right-to-left shunt in migraine with aura: a survey on 120 Italian patients. Neurol. Sci. 30(Suppl. 1), S109–S111 (2009).
- Anzola Gp, Meneghetti G, Zanferrari C, Adami A, Dinia L, Del Sette M: Is migraine associated with right-to-left shunt a separate disease? Results of the sam study. Cephalalgia 28(4), 360–366 (2008).
- Domitrz I, Mieszkowski J, Kaminska A: Relationship between migraine and patent foramen ovale: a study of 121 patients with migraine. Headache 47(9), 1311–1318 (2007).
- Tembl J, Lago A, Sevilla T, Solis P, Vilchez J: Migraine, patent foramen ovale and migraine triggers. J. Headache Pain 8(1), 7–12 (2007).
- Carod-Artal Fj, Da Silveira Ribeiro L, Braga H, Kummer W, Mesquita Hm, Vargas Ap: Prevalence of patent foramen ovale in migraine patients with and without aura compared with stroke patients. A transcranial doppler study. Cephalalgia 26(8), 934–939 (2006).
- Dalla Volta G, Guindani M, Zavarise P, Griffini S, Pezzini A, Padovani A: Prevalence of patent foramen ovale in a large series of patients with migraine with aura, migraine without aura and cluster headache, and relationship with clinical phenotype. J. Headache Pain 6(4), 328–330 (2005).
- Morandi E, Anzola Gp, Angeli S, Melzi G, Onorato E: Transcatheter closure of patent foramen ovale: a new migraine treatment? J. Interv. Cardiol. 16(1), 39–42 (2003).
- Post Mc, Thijs V, Herroelen L, Budts W: Closure of a patent foramen ovale is associated with a decrease in prevalence of migraine. Neurology 62(8), 1439–1440 (2004).
- Azarbal B, Tobis J, Suh W, Chan V, Dao C, Gaster R: Association of interatrial shunts and migraine headaches: impact of transcatheter closure. J. Am. Coll. Cardiol. 45(4), 489–492 (2005).
- Reisman M, Christofferson Rd, Jesurum J et al.: Migraine headache relief after transcatheter closure of patent foramen ovale. J. Am. Coll. Cardiol. 45(4), 493–495 (2005).
- Mortelmans K, Post M, Thijs V, Herroelen L, Budts W: The influence of percutaneous atrial septal defect closure on the occurrence of migraine. Eur. Heart J. 26(15), 1533–1537 (2005).
- Rigatelli G, Cardaioli P, Giordan M et al.: Transcatheter interatrial shunt closure as a cure for migraine: can it be justified by paradoxical embolism-risk-driven criteria? Am. J. Med. Sci. 337(3), 179–181 (2009).
- Kruit Mc, Launer Lj, Ferrari Md, Van Buchem Ma: Infarcts in the posterior circulation territory in migraine. The population-based MRI camera study. Brain 128(Pt 9), 2068–2077 (2005).
- Peto R, Gray R, Collins R et al.: Randomised trial of prophylactic daily aspirin in british male doctors. Br. Med. J. (Clin. Res. Ed.) 296(6618), 313–316 (1988).
- Buring Je, Peto R, Hennekens Ch: Low-dose aspirin for migraine prophylaxis. JAMA 264(13), 1711–1713 (1990).
- Rahimtoola H, Egberts Ac, Buurma H, Tijssen Cc, Leufkens Hg: Reduction in the intensity of abortive migraine drug use during coumarin therapy. Headache 41(8), 768–773 (2001).
- Fragoso Yd: Reduction of migraine attacks during the use of warfarin. Headache 37(10), 667–668 (1997).
- Sharifi M, Dehghani M, Mehdipour M, Al-Bustami O, Emrani F, Burks J: Intense migraines secondary to percutaneous closure of atrial septal defects. J. Interv. Cardiol. 18(3), 181–183 (2005).
- Wilmshurst Pt, Nightingale S, Walsh Kp, Morrison Wl: Clopidogrel reduces migraine with aura after transcatheter closure of persistent foramen ovale and atrial septal defects. Heart 91(9), 1173–1175 (2005).
- Meier B: Stroke and migraine: a cardiologist’s headache. Heart 95(7), 595–602 (2009).
▪ One of the first reports to document an increased prevalence of right-to-left shunts in patients with migraine with aura.
Mainly divers undergoing patent foramen ovale (PFO) closure for prevention of recurrent decompression illness events reported an unexpected reduction of migraine after shunt closure. This report was the first of its kind.
▪ Up-to-date review of the epidemiological data regarding stroke risk in migraine patients. Unfortunately, the presence of a PFO is not yet seriously taken into account.
▪ The only study so far using transesophageal echocardiography in migraine patients to characterize the size and anatomy of intracardiac shunts.
▪ An elegant study that nicely demonstrates the increased shunt size in migraineurs compared with stroke patients and controls.
▪ An interesting attempt to quantify the available evidence regarding the assocation of a PFO and migraine, as well as the effects of shunt closure on migraine.
▪ To date, the only randomized controlled trial assessing the efficacy of percutaneous shunt closure on migraine, albeit with important limitations.
▪ An instructive summary of the ongoing controversies over the conduct and conclusions of the Migraine Intervention with STARflex® Technology (MIST) trial.