Review Article - Imaging in Medicine (2011) Volume 3, Issue 1
New applications of imaging techniques for monitoring progression of rheumatoid arthritis and predicting outcome
Paul Bird†1 and Fredrick Joshua1
1University of New South Wales, Sydney,NSW, Australia
- *Corresponding Author:
- Paul Bird
University of New South Wales Sydney
NSW, Australia
E-mail: pcbird@bigpond.net.au
Abstract
Imaging continues to assume an increasingly important role in the assessment of patients with rheumatoid arthritis. Application of MRI and Doppler ultrasound to this group of patients has revealed important insights into disease processes in rheumatoid arthritis, identifying previously concealed processes that cannot be viewed with conventional imaging. These processes include osteitis, synovitis and early bone erosion. Together, these imaging techniques provide methods for assessment of treatment response as well as short- and long-term outcome. This article examines the current state of imaging in rheumatoid arthritis, with an emphasis on assessment of treatment outcome.
Keywords
bone erosion; Doppler ultrasound; MRI; osteitis; rheumatoid arthritis; synovitis
Rheumatoid arthritis (RA) is a chronic multisystem disorder that affects 1% of the adult population, causing considerable morbidity, a reduction in life expectancy and a significant burden to the community in terms of economic cost [1]. Untreated, patients with RA have a poor prognosis, with the major cause of death being cardiovascular disease [2,3].
The ultimate goals of therapy in RA are to prevent or control joint activity and damage, thereby preventing loss of function and disability [4], and reducing cardiovascular events [5,6]. With treatment options expanding and treatment regimens becoming more complex, the search for precise methods to measure treatment objectives is becoming more urgent. As new therapies emerge, rheumatologists must be able to assess rapidly whether the new treatment can achieve the target objectives better than an older, perhaps safer alternative, and thereby whether the risks of exposure to new or multiple medications justifies the risk to the patient and the cost to the community.
Medical imaging is becoming increasingly important in assessing outcome in patients with RA. Combined with clinical and laboratory techniques, imaging in RA represents a powerful tool for the assessment of outcome.
This article will examine the important role of imaging, incorporating radiographs, CT Doppler ultrasound and MRI, in the assessment of patients with RA.
Rheumatoid arthritis
Rheumatoid arthritis is a chronic, systemic inflammatory disorder with unknown etiology. Joint manifestations are accompanied by elevation of the inflammatory parameters, such as the erythrocyte sedimentation rate and the C-reactive protein, as well as anemia of chronic disease. The disease predominantly involves joints in a symmetrical, polyarticular fashion and leads to joint pain and swelling. The clinical course is variable, but in many cases, the disease causes joint destruction, deformity and disability.
The diagnosis is based upon a total score of 6 or greater (of a possible 10) from the individual scores in four domains from the American College of Rheumatology (ACR)/ The European League Against Rheumatism (EULAR) Criteria [7], present for 6 or more weeks:
• Number and site of involved joints (range 0–5)
• Serological abnormality (range 0–3)
• Elevated acute-phase response (range 0–1)
• Symptom duration (two levels; range 0–1)
In addition to the joint manifestations, the systemic nature of the disease is reflected in constitutional features, such as fatigue, lethargy, weight loss and, occasionally, fevers. The prevalence of RA differs throughout the world, being less in developed rural areas. The most recent prevalence estimates from the Rochester study are 1.4% for women and 0.74% for men [8]. This finding is supported by other studies [9–12] of prevalence in developed populations, leading to the generally quoted figure of prevalence between 0.5 and 1% of the adult population [13].
The inflamed synovium is considered to be the central player in the pathogenesis of RA and although synovitis and bony damage seem to be intertwined, more recent evidence suggests that additional factors may be important in bone destruction in RA [14,15].
Treatment
There has been a dramatic shift in the approach to treatment of RA in the past 10 years.
A major advance has been the introduction of biological agents, notably etanercept, adalimumab and infliximab, which have demonstrated efficacy in reducing disease activity and have the capacity to slow damage progression [16–19]. The chief advantage of these agents is their rapid onset of action compared with traditional disease-modifying antirheumatic drugs, providing symptom relief in weeks rather than months [20], and the capacity to slow damage progression alone or in combination with methotrexate [21,22]. Further agents targeting Bâ�?�?cell receptors [23], Tâ�?�?cell receptors [24] and ILâ�?�?6 [25] are available, expanding the therapeutic armamentarium for treatment. Treatment has shifted dramatically, from an observational approach, to an aggressive multidrug intervention, treating patients to normalize inflammatory parameters and clinical indices [26].
What is clear from the preceding discussion is that treatment for RA is more complex than it has ever been. The obligation of the rheumatologist is to decide which therapy will produce the most beneficial effect for the least toxicity. To perform this function, the rheumatologist and the researcher need a sensitive instrument to document synovitis and detect joint damage progression early, with recordable change in weeks or months, to allow informed decisions regarding changes in therapy.
Imaging provides an opportunity to assist in fulfilling this important objective. The next part of this article will examine the three main methods utilized for assessment of patients with RA – radiographs, ultrasound and MRI – and their respective roles in the assessment of patients with RA.
Methods of imaging assessment in RA
Radiographs
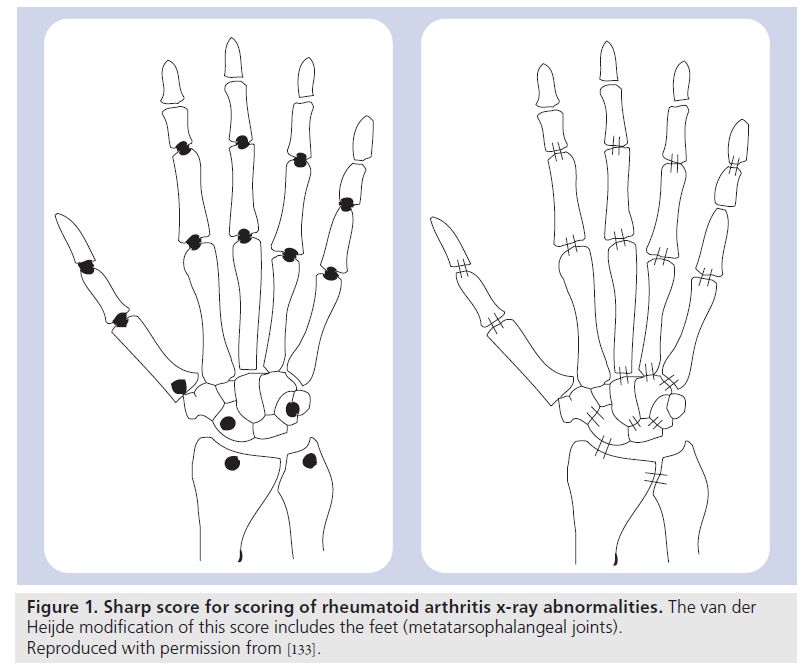
Radiographic imaging is widely accepted as the gold standard for the assessment of disease progression in RA. The major radiographic indicators of damage in the hands and feet of patients with early RA are joint space narrowing, periarticular osteoporosis and bony erosions [27]. Radiographic scores, such as the Larsen and Sharp scores [28,29], and their modifications [30–32], are the standard methods to determine joint damage and its progression. The Sharp score evaluates joint erosion with a score between 0 and 5 for each joint, with a separate score for joint space narrowing on a scale of 0–4 per joint. The Larsen score assesses the degree of joint destruction with a single score from 0 to 5 – the score is mainly determined by erosive changes [33]. The original scoring methods included only the hands, but later modifications of the Sharp score have included the feet as part of the scoring method (Figure 1).
Figure 1. Sharp score for scoring of rheumatoid arthritis x-ray abnormalities. The van der Heijde modification of this score includes the feet (metatarsophalangeal joints). Reproduced with permission from [133].
The advantages of radiographs are that they provide an easily accessible, low-cost, permanent record of joint damage in patients with RA [34]. Reproducibility of the scoring methods has been demonstrated over a broad spectrum of severity (intra-reader and inter-reader) [35,36] as well as sensitivity to change in longitudinal studies [37], particularly for the Sharp method [38].
Despite these documented benefits, radiographs have significant limitations. In RA, these limitations relate primarily to sensitivity, especially in early disease, and responsiveness, both in clinical practice and in drug trials. These factors in turn are affected by image acquisition specifications, by reader variability, and also by the phenomenon of ceiling effects, especially in patients with more advanced disease [39].
The issue of sensitivity is an understandable one. Radiographs are a 1D method of representing complex 3D structures and whilst providing excellent spatial resolution, impart poor contrast resolution when compared with techniques such as CT or MRI. The result is that pathology is sometimes difficult to distinguish from normal anatomy, especially in early disease, with the consequence that there is a well-documented lag time of up to 12 months after diagnosis before definite abnormalities become apparent [40,41]. This has implications not only for clinical practice but also for drug trials in patients with newly diagnosed RA.
The issue of contrast resolution also has an impact on the responsiveness of the radiographs – the fundamental property of providing an adequate representation of damage progression. Radiographs have an impressive track record in documenting damage progression in clinical trials of new therapeutic agents and remain the gold standard in this respect [42], but the framework is shifting rapidly.
This is reflected in the lack of correlation between radiographic findings and measures of disability in patients with RA in clinical trials [43]. With respect to longer term cohorts Scott et al. have demonstrated that there is a poor correlation between radiographic damage and loss of physical function until disease has reached 8–15 years [44]. This lack of correlation confounds attempts to determine a meaning for the important minimal difference in radiographic progression and raises concerns that radiographs may represent only the tip of the joint damage iceberg.
The other major drawback of radiographs is that they provide only a qualitative, rather than a quantitative, expression of joint damage and joint damage progression. To circumvent these problems, quantitative radiographic analysis has been examined by a number of groups. Inexpensive methods have included erosion volume and joint space measurement using plastic template overlays [45]. This technique demonstrated good correlation with standard Sharp scores but reader reliability was not assessed. More recently, computerized image analysis of joint space and erosion volumes in the hands have been trialed by several groups. The computerized system has proved to be reproducible on each occasion, but showed poor correlation with standard joint space scoring in the study by Angwin et al. [46], and poor correlation between erosion volume and the standard erosion score in the study by Sharp et al. [47].
Therefore, radiographs remain the gold standard for assessing outcome and damage progression in clinical trials [48], but this is rapidly altering, with MRI and ultrasound providing viable, sensitive and responsive alternatives.
Ultrasound
Ultrasound features that are evaluated in RA for determining disease activity include evaluation of synovitis and tendon inflammation, and in terms of damage, bone erosions. The validity of ultrasound is established through comparison to other methods of imaging (construct validity), as well as to histology (criterion validity). Importantly for the evaluation of synovitis there are also data to support responsiveness to change after the treatment of RA.
Bone erosion
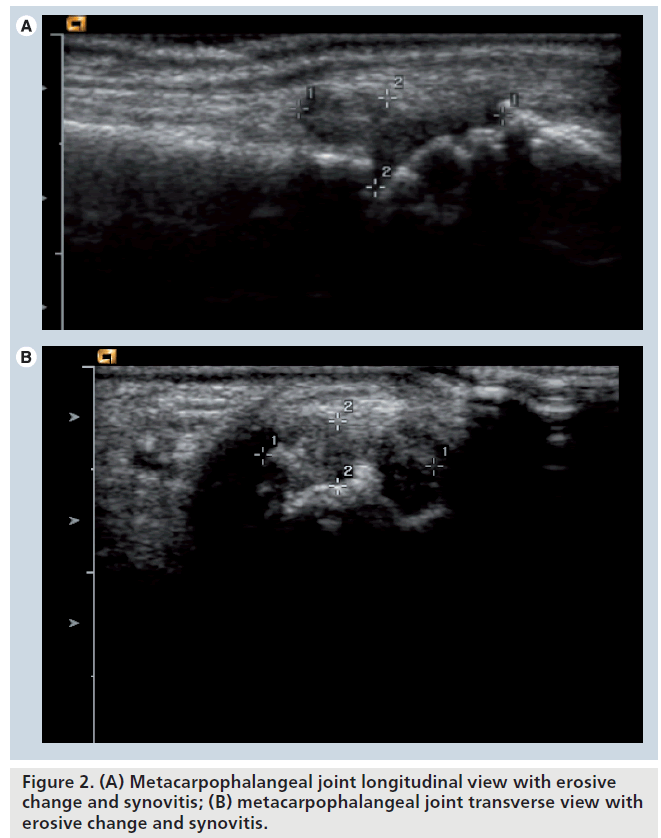
Ultrasound provides information on the bone surface, allowing the evaluation of cortical defects in RA. Bony erosions are an intraarticular discontinuity of the bone surface that is visible in two perpendicular planes [49]. This appears as hypoechoic areas along the surface of hyperechoic bone (Figure 2A & 2B).
The difficulties in ultrasound evaluation of erosions are in two areas. First, the reliability of acquisition of the ultrasound image, and second the inability to insonate past other bony surfaces precludes total evaluation of the 3D structure of the bony surface.
Therefore, in comparison to other imaging techniques the greater definition of the bony surface on ultrasound in comparison to plain radiograph provides advantages, whilst CT and MRI will provide more information given the greater joint coverage. Certainly, in areas where there is good access to the joint, for example at the radial aspect of metacarpophalangeal (MCP)-2, ultrasound is equivalent to MRI in evaluating erosions [50].
The ability of ultrasound to evaluate bone erosions superiorly to plain radiographs but inferiorly to MRI and CT are well described by cross-sectional studies in the metacarpophalangeal joints and metatarsophalaneal joints [51,52]. Longitudinal studies at the MCP joints and wrist have further confirmed that ultrasounddetected erosions are seen on plain radiographs at 7 years [53]. Importantly, ultrasound-detected erosion are shown to reflect erosions seen on CT and MRI, confirming the validity of ultrasound erosion evaluation [54]. Further ultrasound is more responsive to erosions progression over 12 months in early disease when compared with conventional radiology [55,56].
Cartilage
Cartilage can be seen as a hypoechoic layer above the hyperechoic bone on ultrasound. It has not been well studied in RA and further work is progressing to try to establish whether ultrasound evaluation is both valid and practical.
There are increasing data regarding the use of ultrasound in the evaluation of cartilage in disorders such as osteoarthritis and crystal arthropathies, but discussion of these entities is beyond the scope of this article.
Tendon pathology
The available data for tendon evaluation in RA are somewhat heterogeneous in terms of results, and this section therefore reflects the available data.
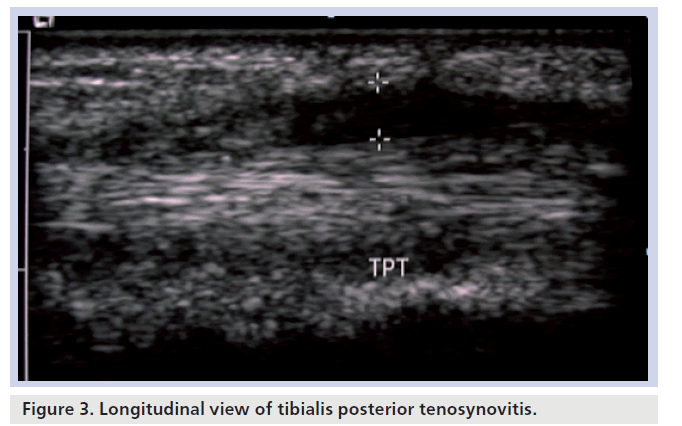
Tenosynovitis in RA can be depicted on ultrasound as thickening of the tendon with a hypoechoic layer surrounding the tendon. There is often Doppler flow in the tendon sheath in association with the hypoechoic fluid (Figure 3). Ultrasound in comparison to MRI in evaluation of tenosynovitis in limited studies suggests similar to slightly better results with MRI [57,58]. Tendon tears appear as a hypoechoic defect in the fibrillar pattern of the tendon on ultrasound. Similar results to the limited tenosynovitis studies comparing ultrasound to MRI were seen in the detection of tendon tears in RA [59].
Synovitis
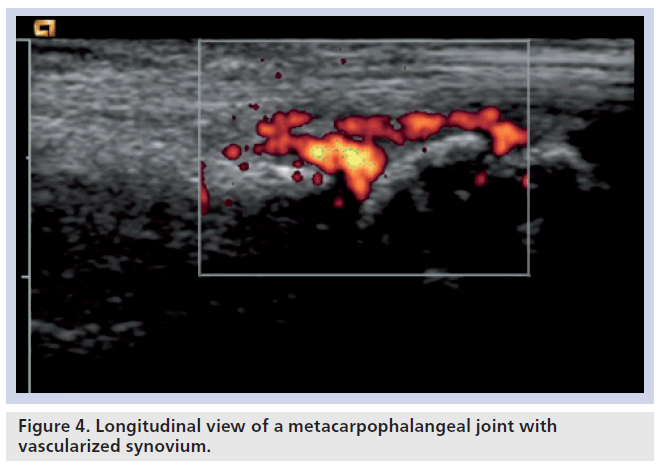
This is the most studied area with ultrasound in RA. It is seen on B-mode ultrasound as a hypoechoic area, which is often hyperemic with Doppler sonography (Figure 4). Technically, power Doppler is the best method of detecting hyperemia of synovium in comparison with color Doppler, as it has in general a higher sensitivity in low blood flow tissues. It is also a more accurate depiction of the number of red blood cells in comparison with the speed and direction of blood flow that is shown with color Doppler sonography [60,61].
Validation of ultrasound-detected synovitis has been performed against histology in the knee and hip and, importantly, in these studies the Doppler signal was shown to correlate with histological vessel numbers [62]. Ultrasound detection of synovitis has also been evaluated against MRI in the knee, wrist, metacarpophalangeal, metatarsophalangeal joints, ankle and shoulder, all showing the superiority for the detection of synovitis with MRI in comparison with ultrasound related to the better total joint capture of MRI [63–65].
There are multiple studies showing that synovitis on ultrasound reduces in size and vascularity with treatment [66–68]. Importantly, the presence of vascular synovium seen on ultrasound has been shown to predict erosive change [69]. This relationship has been seen in other studies confirming the predictive validity of ultrasound [70,71]. The work by Brown et al., in particular, has shown that the presence of ultrasound-detected synovitis predicts erosive change in RA patients thought to be in clinical remission [71].
Scoring the degree of synovitis and vascularity has not been standardized, although there are a number of semiquantitive scoring systems, usually grading from 0 to 3, or none, mild, moderate and severe [72–74]. Quantitative scoring systems use the amount of synovium on grayscale through measurement in a single axis, but this has been difficult due to reliability issues. The quantification of Doppler results has been performed through outlining a region of interest and either measuring the number of color pixels or the color pixel intensity [75,76]. The OMERACT ultrasound group is moving towards a global scoring system that is still in development but has undergone the initial stages of reliability testing [77].
MRI
Several MRI outcome features have emerged as most frequently utilized for the assessment of RA. These were measures of activity (e.g., synovial assessment, bone edema [osteitis] and tendon inflammation) and measures of damage (e.g., bone erosions and cartilage loss) [78]. First, a brief discussion of each MRI abnormality will be presented. It is important to recognize that the criterion validity of the MRI lesions has not been completely established; therefore, the appearance of each abnormality is discussed with the relevant information regarding validity for each finding.
Bone erosion
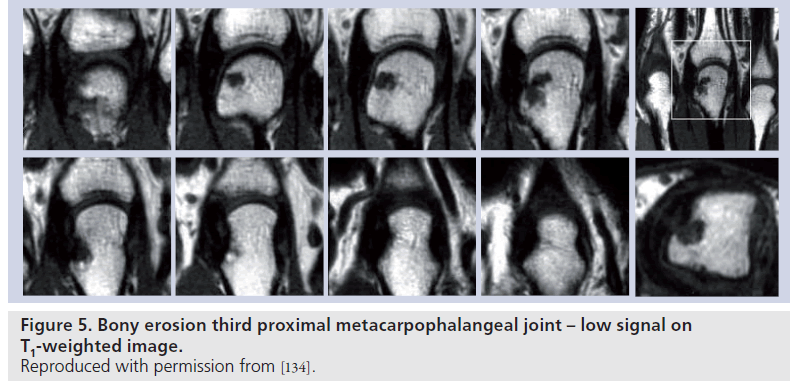
MRI provides 3D representation of bony structures, and therefore provides better definition of bony abnormalities when compared with plain radiography, especially in complex regions, such as the wrist. Bone lesions regarded as bony erosions on MRI appear as focal areas of reduced signal [79] intensity on T1-weighted images (Figure 5).
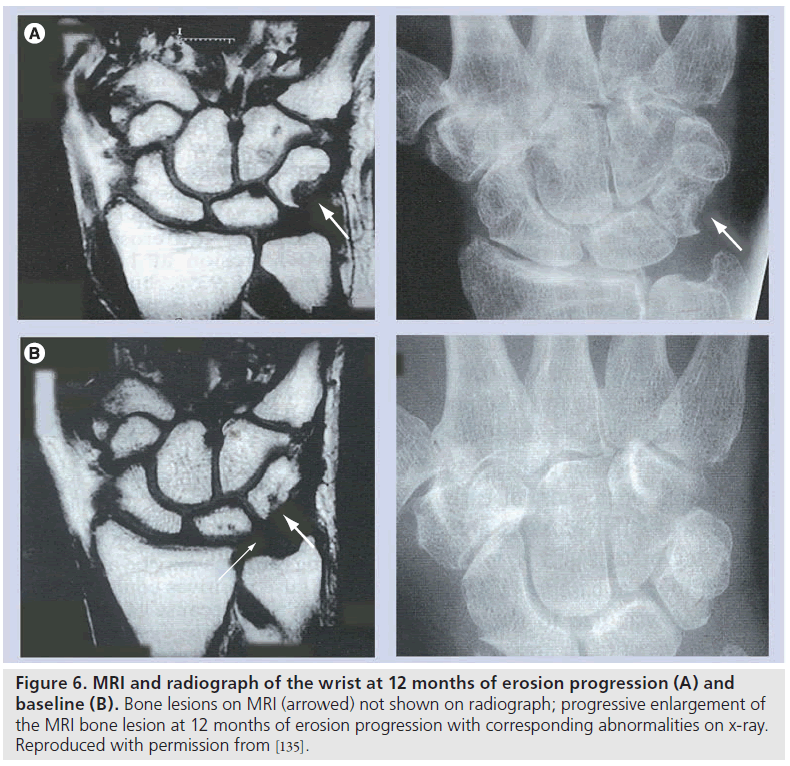
The advantages of MRI of the wrist and MCP joints when compared with radiographs are well documented in cross-sectional studies of patients with early disease (disease duration less than 12 months) [80–85]. MRI provides earlier identification of erosions, with MRI abnormalities appearing well before radiographic change. Importantly, longitudinal studies of unilateraldominant wrist MRI, compared with standard radiographs in early disease, have shown that MRI is more responsive to erosion progression over a 12â�?�?month period [86] and that early MRI erosions correspond to radiographic erosions appearing at 2 and 6 years (Figure 6) [87,88].
Figure 6.MRI and radiograph of the wrist at 12 months of erosion progression (A) and baseline (B). Bone lesions on MRI (arrowed) not shown on radiograph; progressive enlargement of the MRI bone lesion at 12 months of erosion progression with corresponding abnormalities on x-ray. Reproduced with permission from [135].
The issue of joint capture was an early barrier for MRI. While radiographs are not responsive in the short term to joint damage progression, they do provide capture of more joints than MRI (both hands and feet). However, rigorous examination of this issue demonstrated that MRI of the MCP joints and wrist of the dominant hand is equivalent to radiographs of the hands and feet in documenting RA progression [89,90]. What MRI loses in joint capture, it gains in responsiveness and, therefore, MRI of the dominant wrist and MCP joints can be viewed as an adequate representation of global joint damage progression.
Osteitis (bone edema)
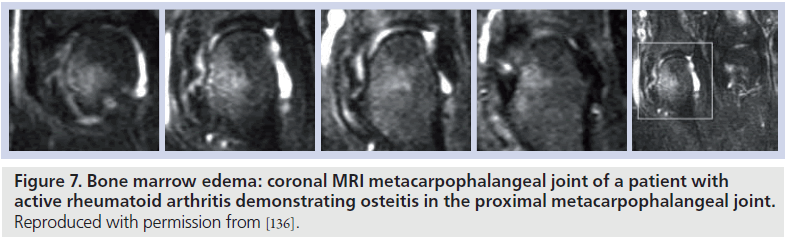
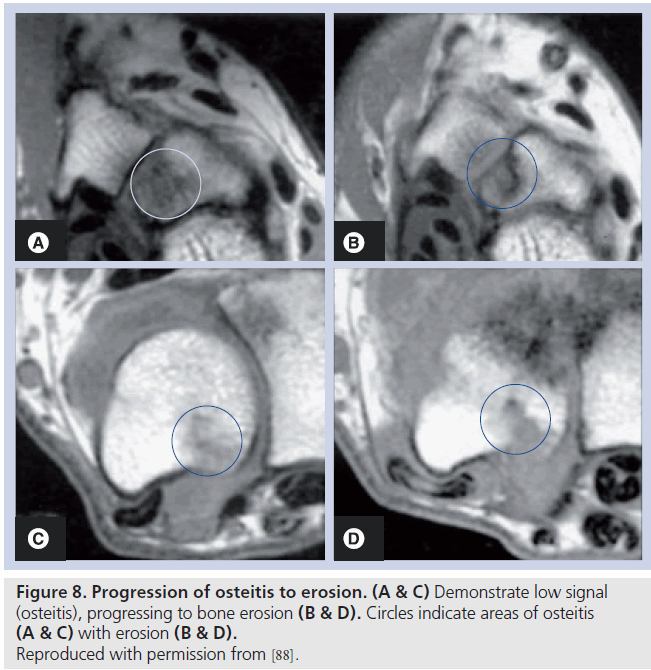
Areas of bone abnormality, termed bone edema, appear as areas of low signal intensity on T1-weighted images, with high signal intensity apparent on corresponding T2-weighted images. The lesion is often described as ‘feathery’ in appearance, with ill-defined margins (Figure 7). In practice, fat-suppressed T2-weighted images are employed to identify areas of bone marrow edema, providing a more distinct delineation of the bone marrow pathology [91]. There is strong evidence to suggest that bone marrow edema precedes the development of subsequent MRI and radiographic erosion [92,93], and therefore the finding has been proposed as a predictor for erosion in patients with early RA (Figure 8) [94,95]. The finding has been demonstrated in surgical specimens of patients with RA [96] undergoing joint replacement, and increased numbers of osteoclasts and RANK-ligand have been demonstrated on pathological specimens. Osteoclast numbers and expression of RANK-ligand, two important factors in bone erosion, have been noted in higher concentration in the areas on MRI osteitis [97].
Figure 7.Bone marrow edema: coronal MRI metacarpophalangeal joint of a patient with active rheumatoid arthritis demonstrating osteitis in the proximal metacarpophalangeal joint. Reproduced with permission from [136].
Osteitis represents a special issue. In RA, some areas of osteitis progress to erosion, but the association is not absolute, and there is evidence that areas of osteitis can regress, being ephemeral in nature, and not leading to erosion. For example, the study by McQueen et al. demonstrated a positive relationship between osteitis and erosion, but the relationship was modest, indicating that whilst it is a reasonable predictor of subsequent damage, not all osteitis leads to erosion [98]. The sensitivity and specificity of osteitis in RA has not been established, and this remains an ongoing research issue. The challenge for researchers is to identify what MRI features are candidates to allow discrimination between nonthreatening and threatening areas of osteitis.
Synovitis
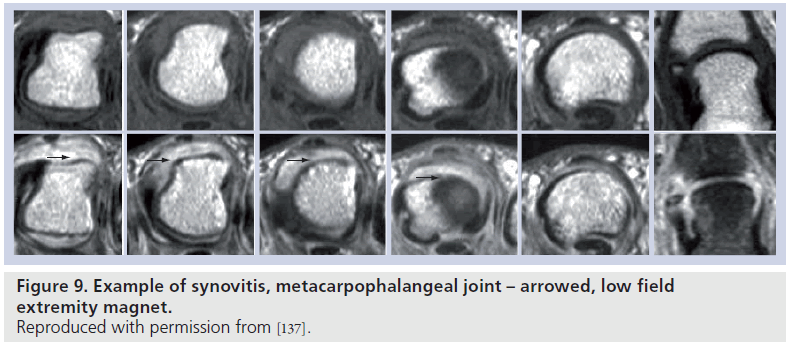
Normal synovial tissue does not enhance postgadolinium – synovitis appears as an area of increased signal intensity greater than the width of normal synovium, on T1-weighted postgadolinium images (Figure 9) [99]. Synovitis can be detected by static or dynamic methods. Obviously, the synovial tissue cannot be visualized by radiographs and study of the appearance on imaging of the synovial membrane in patients with RA is restricted to MRI and ultrasound.
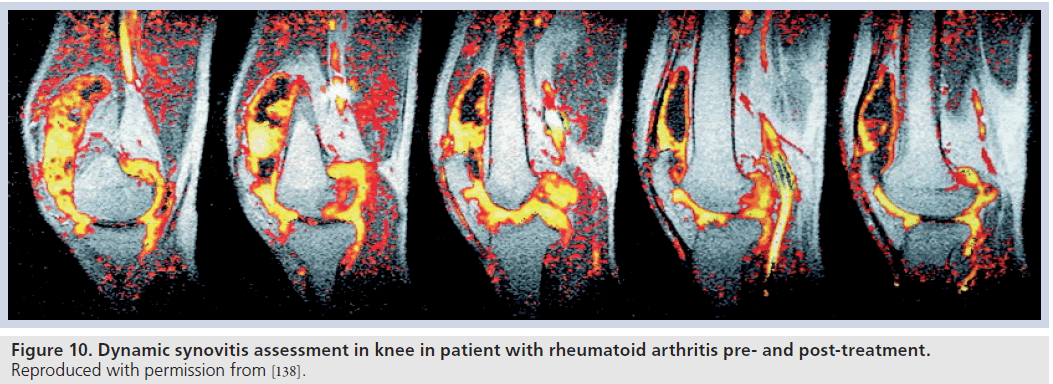
Synovitis has been correlated with subsequent erosive disease on MRI [100,101] and has been used as an indicator of treatment efficacy (Figure 10) [102–104].
Figure 10.Dynamic synovitis assessment in knee in patient with rheumatoid arthritis pre- and post-treatment. Reproduced with permission from [138].
Validation of the MRI abnormality identified as synovitis has been more rigorously evaluated for the knee and MCP joints.
Tendon pathology
Tendonitis, tendon sheath inflammation and tendon rupture are all depicted by MRI [105]. While MRI findings correlate with clinical and ultrasound findings [106,107], the significance of tendon pathology in predicting erosive disease is uncertain, as only one group [108] have incorporated tendon abnormalities as part of a global scoring system. Therefore, the significance of tendon abnormalities in RA remains an ongoing research issue.
Cartilage defects/cartilage loss
A standard T1 axial and coronal sequence is accepted widely as providing adequate resolution for the detection of erosions, but this does not provide adequate visualization of cartilage. In larger joints, such as the knee, 3D gradient echo has been recommended as the most effective sequence for estimating cartilage defects and cartilage volume in osteoarthritis [109]. Unfortunately, studies examining the role of MRI in detecting cartilage defects in the knee in RA are small in number [110–114] without clear validation, and this remains an area where there is a lack of information regarding one of the important processes in RA. The visualization of cartilage in the MCP joints and wrist is an even more difficult problem – whilst 3D gradient echo sequences provide some detail in this region, there are still problems related to the size of the joints in the hand, leading to problems differentiating articular cartilage from underlying bone. The use of thin partition 3D MRI combined with T1-weighted images and specialized surface coils has been shown to be advantageous in delineating cartilage in the MCP joints [115], but studies examining the reproducibility of this method in large numbers of patients with RA are lacking. The OMERACT group is pursuing a cartilage score, but at the time of writing, the score has not been published.
MRI measurement technique OMERACT MRI score
In 2003, the OMERACT [116] group published an international scoring system – a system with documented inter- and intra-reader reliability with a semiquantitative approach to scoring erosions [117]. Through rigorous evaluation, reproducibility of the score for synovitis, osteitis and bone erosion has been established for both cross-sectional and longitudinal studies [118,119].
In 2004 an atlas was published, providing reference images and score rationalization [120,121]. This scoring system has become the standard method for scoring MRI in clinical trials, replacing previous scoring systems.
Quantitative measurement
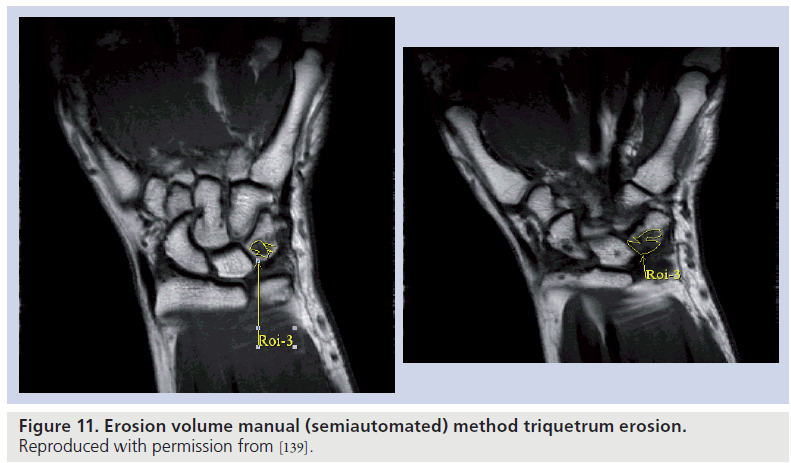
Erosion volume and synovial volume measurement has been undertaken by several groups [122–124], with the aim of providing a more accurate representation of joint damage and inflammatory activity. Semiautomated and automated computer programs have been utilized, with correlation demonstrated between erosion volumes and the OMERACT erosion score (Figure 11). Barriers to widespread use largely relate to the time required for segmentation, and the lack of available software to provide a truly automated volume in joints. The recent development of software designed to display dynamic 3D images [125] will assist in the pursuit of automated volumes, and this remains an important goal for researchers.
Computerized tomography
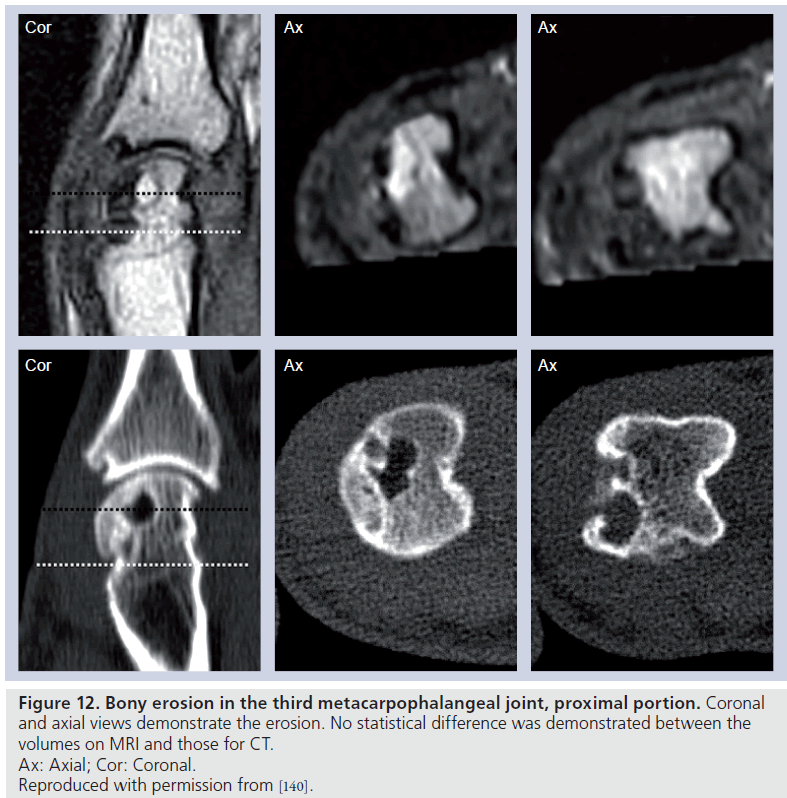
Computerized tomography is arguably the best method to demonstrate bony pathology, and entails use of ionizing radiation. This is not ideal, especially in situations where repeat examinations are required in a short period of time. The main utility of CT is related to erosion detection, but the technique does not provide information regarding synovitis and osteitis. Comparison studies of CT and MRI [126–128] in the assessment of bone erosion in patients with RA have shown no significant difference for detection of erosions or measurement of erosion size (Figure 12). Therefore, while CT remains a possible technique for assessment of RA patients, limitations of information gained with each examination, as well as concerns regarding ionizing radiation, have limited its widespread application.
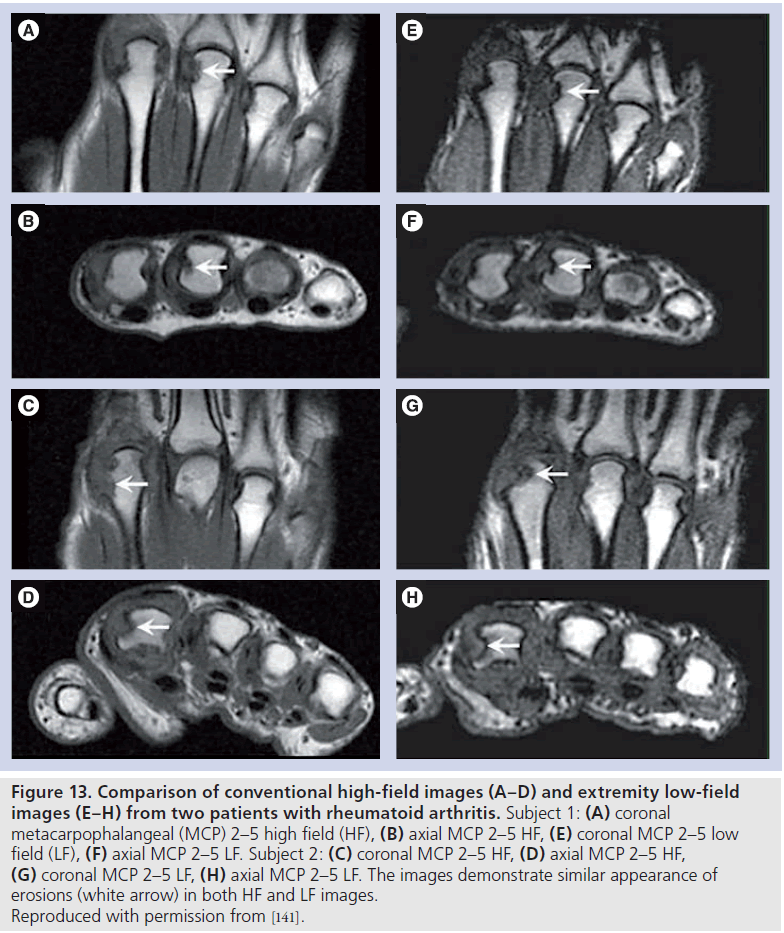
Low-field versus high-field MRI comparison
Extremity MRI is an attractive alternative to conventional MRI; the main advantages of this method over conventional MRI include lower cost, enhanced patient comfort and reduced imaging time. The main perceived disadvantage of the low-field method is lower signal-tonoise ratio, which may affect image quality, and therefore erosion, osteitis and synovitis scoring.
Initial unireader studies comparing low-field and high-field MRI [129,130] in RA demonstrated an advantage of high-field over low-field MRI. More recent multireader studies have demonstrated excellent inter-reader agreement for erosions, with the apparent lower image quality of extremity imaging not affecting erosion scoring (Figure 13) [131]. Osteitis and synovitis agreement was, however, less robust in these studies, but still demonstrated acceptable inter-reader reliability. Whilst this is not a barrier to the use of low-field extremity imaging in the assessment of synovitis and osteitis, caution needs to be exercised when comparing low- and high-field measures of activity, particularly if readers are not familiar with the appearance of synovitis and osteitis on low-field imaging [132].
Figure 13Comparison of conventional high-field images (A–D) and extremity low-field images (E–H) from two patients with rheumatoid arthritis. Subject 1: (A) coronal metacarpophalangeal (MCP) 2–5 high field (HF), (B) axial MCP 2–5 HF, (E) coronal MCP 2–5 low field (LF), (F) axial MCP 2–5 LF. Subject 2: (C) coronal MCP 2–5 HF, (D) axial MCP 2–5 HF, (G) coronal MCP 2–5 LF, (H) axial MCP 2–5 LF. The images demonstrate similar appearance of erosions (white arrow) in both HF and LF images. Reproduced with permission from [141].
Future perspective
MRI and ultrasound have an increasing role in the assessment of outcome in patients with RA. Both techniques have provided important insights into disease pathogenesis, providing examination of structures not previously available for scrutiny with plain radiographs. Both techniques provide methods for early identification of disease, and because both techniques are sensitive, reliable and responsive, they provide an important method for assessing outcome.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Papers of special note have been highlighted as:
*of interest
* of considerable interest
References
- Maetzel A, Li L, Pencharz J, Tomlinson G, Bombardier C: The economic burden associated with osteoarthritis, rheumatoid arthritis, and hypertension: a comparative study. Ann. Rheum. Dis. 63(4), 395–401 (2004).
- Naranjo A, Sokka T, Descalzo MA et al.; QUEST-RA Group: Cardiovascular disease in patients with rheumatoid arthritis: results from the QUEST-RA study. Arthritis Res. Ther. 10(2), R30 (2008).
- Gabriel SE: Cardiovascular morbidity and mortality in rheumatoid arthritis. Am. J. Med. 121(10 Suppl. 1), S9–S14 (2008).
- Kwoh K, Anderson L, Greene J et al.: Guidelines for the management of rheumatoid arthritis. 2002 update. American College of Rheumatology Subcommittee on Rheumatoid Arthritis Guidelines. Arthritis Rheum. 46(2), 328–346 (2002).
- Jacobsson LTH, Turesson C, Gülfe A et al.: Treatment with tumor necrosis factor blockers is associated with a lower incidence of first cardiovascular events in patients with rheumatoid arthritis. J. Rheumatol. 32(7), 1213 (2005).
- Dixon WG, Watson KD, Lunt M et al.: Reduction in the incidence of myocardial infarction in patients with rheumatoid arthritis who respond to anti-tumor necrosis factor a therapy: results from the British Society for Rheumatology Biologics Register. Arthritis Rheum. 56(9), 2905–2912 (2007).
- Aletaha D, Neogi T, Silman A et al.: 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/ European League Against Rheumatism collaborative initiative. Arthritis Rheum. 62(9), 2569–2581 (2010).
- Gabriel SE, Crowson CS, O’Fallon WM: The epidemiology of rheumatoid arthritis in Rochester, MN 1955–1985. Arthritis Rheum. 42, 415–420 (1999).
- Cimmino MA: Prevalence of rheumatoid arthritis in Italy: the Chiavari study. Ann. Rheum. Dis. 57, 315–318 (1998).
- Boyer GS, Benvolenskaya LI, Templin DW et al.: Prevalence of rheumatoid arthritis in circumpolar native populations. J. Rheumatol. 25, 23–29 (1998).
- Drosos AA, Alamanos I, Voulgari PV et al.: Epidemiology of adult rheumatoid arthritis in northwest Greece 1987–1995. J. Rheumatol. 24, 2129–2133 (1997).
- Aho K, Kaipiainen O, Heliovaara M et al.: Epidemiology of rheumatoid arthritis in Finland. Semin. Arthritis Rheum. 27, 325–344 (1998).
- Symmons DP: Epidemiology of rheumatoid arthritis: determinants of onset, persistence and outcome. Best Pract. Res. Clin. Rheumatol. 16(5), 707–722 (2002).
- Kirwan J:Links between radiological change, disability, and pathology in rheumatoid arthritis. J. Rheumatol. 28(4), 881–886 (2001).
- Cunnane G, FitzGerald O, Hummel KM et al.: Synovial tissue protease gene expression and joint erosions in early rheumatoid arthritis. Arthritis Rheum. 44(8), 1744–1753 (2001).
- Weinblatt ME, Kremer JM, Bankhurst AD et al.: A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N. Eng. J. Med. 340, 253–259 (1999).
- Maini R, St Clair EW, Breedveld F et al.: Infliximab (chimeric anti-tumour necrosis factor a monoclonal antibody) versus placebo in rheumatoid arthritis patients receiving concomitant methotrexate: a randomised Phase III trial. ATTRACT Study Group. Lancet 354(9194), 1932–1939 (1999).
- Lipsky PE, van der Heijde DM, St Clair EW et al.; Anti-Tumor Necrosis Factor Trial in Rheumatoid Arthritis with Concomitant Therapy Study Group: Infliximab and methotrexate in the treatment of rheumatoid arthritis. N. Engl. J. Med. 343(22), 1594–1602 (2000).
- O’Dell J: Therapeutic strategies for rheumatoid arthritis. N. Engl. J. Med. 350(25), 2591–2602 (2004).
- Weisman MH, Moreland LW, Furst DE et al.: Efficacy, pharmacokinetic, and safety assessment of adalimumab, a fully human anti-tumor necrosis factor-a monoclonal antibody, in adults with rheumatoid arthritis receiving concomitant methotrexate: a pilot study. Clin. Ther. 25(6), 1700–1721 (2003).
- Klareskog L, van der Heijde D, de Jager JP et al.; TEMPO (Trial of Etanercept and Methotrexate with Radiographic Patient Outcomes) study investigators: Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: doubleblind randomised controlled trial. Lancet 363(9410), 675–681 (2004).
- Singh JA, Christensen R, Wells GA et al.: Biologics for rheumatoid arthritis: an overview of Cochrane reviews. Cochrane Database Syst. Rev. 4, CD007848 (2009).
- Bagust A, Boland A, Hockenhull J et al.: Rituximab for the treatment of rheumatoid arthritis. Health Technol. Assess. 13(Suppl. 2), 23–29 (2009).
- Maxwell L, Singh JA: Abatacept for rheumatoid arthritis. Cochrane Database Syst. Rev. 4, CD007277 (2009).
- Nishimoto N: Interleukin-6 as a therapeutic target in candidate inflammatory diseases. Pharmacol. Ther. 87(4), 483–487 (2010).
- Smolen JS, Aletaha D, Bijlsma JW et al.; T2T Expert Committee: Treating rheumatoid arthritis to target: recommendations of an international task force. Ann. Rheum. Dis. 69(4), 631–637 (2010).
- Scott DL, Coulton BL, Bacon PA, Popert AJ: Methods of x-ray assessment in rheumatoid arthritis: a re-evaluation. Br. J. Rheumatol. 24 (1), 31–39 (1985).
- Larsen A, Dale K, Eek M: Radiographic evaluation of rheumatoid arthritis and related conditions by standard reference films. Acta Radiologica Diag. 18, 481–491 (1977).
- Sharp JT, Young DY, Bluhm GB et al.: How many joints in the hands and wrists should be included in a score of radiologic abnormalities used to assess rheumatoid arthritis? Arthritis Rheum. 28(12), 1326–1335 (1985).
- Scott D, Laasonen L, Priolo F et al.: The radiological assessment of rheumatoid arthritis. Clin. Exp. Rheum. 15(Suppl. 17), S53–S61 (1997).
- van der Heijde DM: How to read radiographs according to the Sharp/van der Heijde method. J. Rheumatol. 26(3), 743–745 (1999).
- Rau R, Herborn G: A modified version of Larsen’s scoring method to assess radiologic changes in rheumatoid arthritis. J. Rheumatol. 22, 1976–1982 (1995).
- van der Heijde DM: Plain x-rays in rheumatoid arthritis: overview of scoring methods, their reliability and applicability. Baillieres Clin. Rheumatol. 10(3), 435–453 (1996).
- Van der Heijde D: Radiographic imaging: the gold standard for assessment of disease progression in rheumatoid arthritis. Rheumatology 39(Suppl. 1), 9–16 (2000).
- Sharp JT, Bluhm GB, Brook A et al.: Reproducibility of multiple observer scoring Arthritis Rheum. 28(1), 16–24 (1985).
- O’Sullivan M et al.: Precision of Larsen grading of radiographs in assessing progression of rheumatoid arthritis in individual patients. Ann. Rheum. Dis. 49, 286–289 (1990).
- van der Heijde D, Dankert T, Nieman F, Rau R, Boers M: Reliability and sensitivity to change of a simplification of the Sharp/van der Heijde radiological assessment in rheumatoid arthritis. Rheumatology 38, 941–947 (1999).
- Scott D, Laasonen L, Priolo F et al.: The radiological assessment of rheumatoid arthritis. Clin. Exp. Rheum. 15(Suppl. 17), S53–S61 (1997).
- van der Heijde DM: Radiographic imaging: the ‘gold standard’ for assessment of disease progression in rheumatoid arthritis. Rheumatology 39(Suppl. 1), 9–16 (2000).
- Norgaard F: Earliest roentological changes in polyarthritis of the rheumatoid type: rheumatoid arthritis. Radiology 85, 325–329 (1965).
- Brower AC: Use of the radiograph to measure the course of rheumatoid arthritis disease. The gold standard vs fool’s gold. Arthritis Rheum. 33, 316–324 (1990).
- van der Heijde DM: Radiographic imaging: the ‘gold standard’ for assessment of disease progression in rheumatoid arthritis. Rheumatology 39(Suppl. 1), 9–16 (2000).
- Strand V, Sharp J: Radiographic data from recent randomized controlled clinical trials in rheumatoid arthritis. What have we learned? Arthritis Rheum. 48(1), 21–34 (2003).
- Scott D, Pugner K, Kaarela K et al.: The links between joint damage and disability in rheumatoid arthritis. Rheumatology (Oxf.) 39, 122–132 (2000).
- Higgs JB, Smith D, Des Rosier KF, Charlesworth RW Jr: Quantitative measurement of erosion growth and joint space loss in rheumatoid arthritis hand radiographs. J. Rheumatol. 23(2), 265–272 (1996).
- Angwin J, Heald G, Lloyd A, Howland K, Davy M, James MF: Reliability and sensitivity of joint space measurements in hand radiographs using computerized image analysis. J. Rheumatol. 28(8), 1825–1836 (2001).
- Sharp J, Gardner J, Bennett E: Computerbased methods for measuring joint space and estimating erosion volume in the finger and wrist joints of patients with rheumatoid arthritis. Arthritis Rheum. 43(6), 1378–1386 (2000).
- Yazici Y, Sokka T, Pincus T: Radiographic measures to assess patients with rheumatoid arthritis: advantages and limitations. Rheum. Dis. Clin. North Am. 35(4), 723–729, VI (2009).
- Wakefield RJ, Balint PV, Szkudlarek M et al.; OMERACT 7 Special Interest Group: Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J. Rheumatol. 32(12), 2485–2487 (2005).
- Szkudlarek M, Narvestad E, Klarlund M, Court-Payen M, Thomsen HS, Østergaard M: Ultrasonography of the metatarsophalangeal joints in rheumatoid arthritis: comparison with magnetic resonance imaging, conventional radiography, and clinical examination. Arthritis Rheum. 50(7), 2103–2112 (2004).
- Wakefield RJ, Gibbon WW, Conaghan PG et al.: The value of sonography in the detection of bone erosions in patients with rheumatoid arthritis: a comparison with conventional radiography. Arthritis Rheum. 43(12), 2762–2770 (2000).
- Hoving JL, Buchbinder R, Hall S et al.: A comparison of magnetic resonance imaging, sonography, and radiography of the hand in patients with early rheumatoid arthritis. J. Rheumatol. 31(4), 663–675 (2004).
- Scheel AK, Hermann KG, Ohrndorf S et al.: Prospective 7 year follow up imaging study comparing radiography, ultrasonography, and magnetic resonance imaging in rheumatoid arthritis finger joints. Ann. Rheum. Dis. 65(5), 595–600 (2006).
- Døhn UM, Ejbjerg BJ, Court-Payen M et al.: Are bone erosions detected by magnetic resonance imaging and ultrasonography true erosions? A comparison with computed tomography in rheumatoid arthritis metacarpophalangeal joints. Arthritis Res. Ther. 8(4), R110 (2006).
- Bajaj S, Lopez-Ben R, Oster R, Alarcón GS: Ultrasound detects rapid progression of erosive disease in early rheumatoid arthritis: a prospective longitudinal study. Skeletal Radiol. 36(2), 123–128 (2007).
- Hammer HB, Haavardsholm EA, Bøyesen P, Kvien TK: Bone erosions at the distal ulna detected by ultrasonography are associated with structural damage assessed by conventional radiography and MRI: a study of patients with recent onset rheumatoid arthritis. Rheumatology (Oxf.) 48(12), 1530–1532 (2009).
- Hoving JL, Buchbinder R, Hall S et al.: A comparison of magnetic resonance imaging, sonography, and radiography of the hand in patients with early rheumatoid arthritis. Rheumatology 31(4), 663–675 (2004).
- Wakefield RJ, O’Connor PJ, Conaghan PG et al.: Finger tendon disease in untreated early rheumatoid arthritis: a comparison of ultrasound and magnetic resonance imaging. Arthritis Rheum. 57(7), 1158–1164 (2007). & Good paper comparing MRI and ultrasound.
- Swen WA, Jacobs JW, Hubach PC, Klasens JH, Algra PR, Bijlsma JW: Comparison of sonography and magnetic resonance imaging for the diagnosis of partial tears of finger extensor tendons in rheumatoid arthritis. Rheumatology (Oxf.) 39(1), 55–62 (2000).
- Rubin JM, Adler RS, Fowlkes JB et al.: Fractional moving blood volume: estimation with power Doppler US. Radiology 197, 183–190 (1995).
- Walther M, Harms H, Krenn V, Radke S, Kirschner S, Gohlke F: Synovial tissue of the hip at power Doppler US: correlation between vascularity and power Doppler US signal. Radiology 225(1), 225–231 (2002).
- Walther M, Harms H, Krenn V, Radke S, Faehndrich TP, Gohlke F: Correlation of power Doppler sonography with vascularity of the synovial tissue of the knee joint in patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum. 44, 331–338 (2001).
- Beckers C, Jeukens X, Ribbens C et al.: (18) F-FDG PET imaging of rheumatoid knee synovitis correlates with dynamic magnetic resonance and sonographic assessments as well as with the serum level of metalloproteinase-3. Eur. J. Nucl. Med. Mol. Imaging 33(3), 275–280 (2006).
- Wakefield RJ, Freeston JE, O’Connor P et al.: The optimal assessment of the rheumatoid arthritis hindfoot: a comparative study of clinical examination, ultrasound and high field MRI. Ann. Rheum. Dis. 67(12), 1678–1682 (2008). & Very useful study, summarizing key issues related to sensitivity, specificity and responsiveness.
- Szkudlarek M, Narvestad E, Klarlund M, Court-Payen M, Thomsen HS, Østergaard M: Ultrasonography of the metatarsophalangeal joints in rheumatoid arthritis: comparison with magnetic resonance imaging, conventional radiography, and clinical examination. Arthritis Rheum. 50(7), 2103–2112 (2004).
- Newman JS, Laing TJ, McCarthy CJ, Adler RS: Power Doppler sonography of synovitis: assessment of therapeutic response – preliminary observations. Radiology 198, 582–584 (1996).
- Ribbens C, Andre B, Marcelis S et al.: Rheumatoid hand joint synovitis: gray-scale and power Doppler US quantifications following anti-tumor necrosis factor-a treatment: pilot study. Radiology 229(2), 562–569 (2003).
- Taylor PC, Steuer A, Gruber J et al.: Comparison of ultrasonographic assessment of synovitis and joint vascularity with radiographic evaluation in a randomized, placebo-controlled study of infliximab therapy in early rheumatoid arthritis. Arthritis Rheum. 50, 1107–1116 (2004).
- Naredo E, Collado P, Cruz A et al.: Longitudinal power Doppler ultrasonographic assessment of joint inflammatory activity in early rheumatoid arthritis: predictive value in disease activity and radiologic progression. Arthritis Rheum. 57(1), 116–124 (2007).
- Taylor PC, Steuer A, Gruber J et al.: Comparison of ultrasonographic assessment of synovitis and joint vascularity with radiographic evaluation in a randomized, placebo-controlled study of infliximab therapy in early rheumatoid arthritis. Arthritis Rheum. 50(4), 1107–1116 (2004).
- Brown AK, Conaghan PG, Karim Z et al.: An explanation for the apparent dissociation between clinical remission and continued structural deterioration in rheumatoid arthritis. Arthritis Rheum. 58(10), 2958–2967 (2008).
- Newman JS, Laing TJ, McCarthy CJ, Adler RS: Power Doppler sonography of synovitis: assessment of therapeutic response – preliminary observations. Radiology 198, 582–584 (1996).
- Stone M, Bergin D, Whelan B, Maher M, Murray J, McCarthy C: Power Doppler ultrasound assessment of rheumatoid hand synovitis. J. Rheumatol. 28, 1979–1982 (2001).
- Weidekamm C, Koller M, Weber M, Kainberger F: Diagnostic value of highresolution B-mode and Doppler sonography for imaging of hand and finger joints in rheumatoid arthritis. Arthritis Rheum. 48, 325–333 (2003). & Excellent summary of technique of Doppler ultrasound.
- Walther M, Harms H, Krenn V, Radke S, Faehndrich TP, Gohlke F: Correlation of power Doppler sonography with vascularity of the synovial tissue of the knee joint in patients with osteoarthritis and rheumatoid arthritis. Arthritis Rheum. 44, 331–338 (2001).
- Taylor PC, Steuer A, Gruber J et al.: Comparison of ultrasonographic assessment of synovitis and joint vascularity with radiographic evaluation in a randomized, placebo-controlled study of infliximab therapy in early rheumatoid arthritis. Arthritis Rheum. 50, 1107–1116 (2004).
- D’Agostino MA, Conaghan PG, Naredo E et al.: The OMERACT ultrasound task force – advances and priorities. J. Rheumatol. 36(8), 1829–1832 (2009).
- Boesen M, Østergaard M, Cimmino MA, Kubassova O, Jensen KE, Bliddal H: MRI quantification of rheumatoid arthritis: current knowledge and future perspectives. Eur. J. Radiol. 71(2), 189–196 (2009). & Excellent summary of MRI quantification methods.
- McQueen F: Magnetic resonance in early inflammatory arthritis: what is its role? Rheumatology 39, 700–706 (2000).
- McQueen FM, Stewart N, Crabbe J et al.: Magnetic resonance imaging of the wrist in early rheumatoid arthritis reveals a high prevalence of erosions at four months after symptom onset. Ann. Rheum. Dis. 57(6), 350–356 (1998).
- Foley-Nolan D, Stack JP, Ryan M et al.: Magnetic resonance imaging in the assessment of rheumatoid arthritis – a comparison with plain film radiographs. Br. J. Rheumatol. 30 (2), 101–106 (1991).
- Jorgensen C, Cyteval CC, Anaya J, Baron M, Lamarque J, Sany J: Sensitivity of magnetic resonance imaging of the wrist in very early rheumatoid arthritis. Clin. Exp. Rheum. 11, 163–168 (1993).
- Klarlund M, Østergaard M, Jensen K et al.: Magnetic resonance imaging, radiography and scintigraphy of the finger joints: one year follow up of patients with early arthritis. Ann. Rheum. Dis. 59, 521–528 (2000).
- Mcgonagle D, Conaghan P, Wakefield D, Emery P: Imaging the joints in early rheumatoid arthritis. Ballieres Best Pract. Res. Clin. Rheumatol. 15(1), 91–104 (2001).
- Lindegaard H, Vallo J, Horslev-Petersen K, Junker P, Østergaard M: Low field dedicated magnetic resonance imaging in untreated rheumatoid arthritis of recent onset. Ann. Rheum. Dis. 60 (8), 770–776 (2001).
- McQueen FM, Stewart N, Crabbe J et al.: Magnetic resonance imaging of the wrist in early rheumatoid arthritis reveals progression of erosions despite clinical improvement. Ann. Rheum. Dis. 58(3), 156–163 (1999).
- McQueen FM, Benton N, Crabbe J et al.: What is the fate of erosions in early rheumatoid arthritis? Tracking individual lesions using x rays and magnetic resonance imaging over the first two years of disease. Ann. Rheum. Dis. 60(9), 859–868 (2001).
- McQueen F, Benton N, Perry D et al.: Bone edema scored on magnetic resonance imaging scans of the dominant carpus at presentation predicts radiographic joint damage of the hands and feet six years later in patients with rheumatoid arthritis. Arthritis Rheum. 48(7), 1814–1827 (2003).
- Bird P, Kirkham B, Portek I et al.: Documenting damage progression in a two-year longitudinal study of rheumatoid arthritis patients with established disease (the DAMAGE study cohort): is there an advantage in the use of magnetic resonance imaging as compared with plain radiography? Arthritis Rheum. 50(5), 1383–1389 (2004).
- Ejbjerg BJ, Vestergaard A, Jacobsen S, Thomsen HS, Østergaard M: The smallest detectable difference and sensitivity to change of magnetic resonance imaging and radiographic scoring of structural joint damage in rheumatoid arthritis finger, wrist, and toe joints: a comparison of the OMERACT rheumatoid arthritis magnetic resonance imaging score applied to different joint combinations and the Sharp/van der Heijde radiographic score. Arthritis Rheum. 52(8), 2300–2306 (2005).
- Peterfy C: Magnetic resonance imaging in rheumatoid arthritis: current status and future directions. J. Rheumatol. 28, 1134–1142 (2001).
- McQueen FM, Stewart N, Crabbe J et al.: Magnetic resonance imaging of the wrist in early rheumatoid arthritis reveals progression of erosions despite clinical improvement. Ann. Rheum. Dis. 58(3), 156–163 (1999).
- McGonagle D, Conaghan PG, O”Connor P et al.: The relationship between synovitis and bone changes in early untreateed rheumatoid arthritis: a controlled magnetic resonance imaging study. Arthritis Rheum. 42, 1706–1711 (1999).
- Peterfy C: New developments in imaging in rheumatoid arthritis. Curr. Opin. Rheumatol. 15, 288–295 (2003).
- Hetland ML, Ejbjerg B, Hørslev-Petersen K et al.; CIMESTRA study group: MRI bone oedema is the strongest predictor of subsequent radiographic progression in early rheumatoid arthritis. Results from a 2-year randomised controlled trial (CIMESTRA). 68(3), 384–390 (2009). & Examines the issue of osteitis as a predictor of erosive change.
- McQueen FM, Gao A, Østergaard M et al.: High-grade MRI bone oedema is common within the surgical field in rheumatoid arthritis patients undergoing joint replacement and is associated with osteitis in subchondral bone. Ann. Rheum. Dis. 66(12), 1581–1587 (2007).
- Dalbeth N, Smith T, Gray S et al.: Cellular characterisation of magnetic resonance imaging bone oedema in rheumatoid arthritis; implications for pathogenesis of erosive disease. Ann. Rheum. Dis. 68(2), 279–282 (2009).
- McQueen F, Benton N, Perry D et al.: Bone edema scored on magnetic resonance imaging scns of the dominant carpus at presentation predicts radiographic joint damage of the hands and feet six years later in patients with rheumatoid arthritis. Arthritis Rheum. 48(7), 1814–1827 (2003).
- Smith HJ: Contrast enhanced MRI of rheumatic joint disease. Br. J. Rheum. 35(Suppl. 3), 45–47 (1996).
- Østergaard M, Hansen M, Stoltenberg M, Lorenzen I: Quantitative assessment of the synovial membrane in the rheumatoid wrist: an easily obtained MRI score reflects the synovial volume. Br. J. Rheumatol. 35(10), 965–971 (1996).
- Conaghan P, O’Connor P, McGonagle D et al.: Elucidation of the relationship between synovitis and bone damage. Arthritis Rheum. 48(1), 64–71 (2003).
- Reece R, Kraan MC, Radjenovic A, Veale D et al.: Comparative assessment of leflunomide and methotrexate for the treatment of rheumatoid arthritis, by dynamic enhanced magnetic resonance imaging. Arthritis Rheum. 46, 366–372 (2002).
- Kalden-Nemeth D, Grebmeier J, Antoni C, Manger B, Wolf F, Kalden JR: NMR monitoring of rheumatoid arthritis patients receiving anti-TNF-a monoclonal antibody therapy. Rheumatol. Int. 16, 249–255 (1997).
- Østergaard M, Stoltenberg M, Henriksen O, Lorenzen I: Quantitative assessment of synovial inflammation by dynamic gadolinium-enhanced magnetic resonance imaging. A study of the effect of intraarticular methylprednisolone on the rate of early synovial enhancement. Br. J. Rheumatol. 35, 50–59 (1996).
- Peterfy C: New developments in imaging in rheumatoid arthritis. Curr. Opin. Rheumatol. 15, 288–295 (2003).
- Valeri G, Ferrara C, Ercolani P et al.: Tendon involvement in rheumatoid arthritis of the wrist: MRI findings. Skeletal Radiol. 30, 138–143 (2001).
- Backhaus M, Kamradt T, Sandrock D et al.: Arthritis of the finger joints: a comprehensive approach comparing conventional radiography, scintigraphy, ultrasound, and contrast-enhanced magnetic resonance imaging. Arthritis Rheum. 42, 1232–1245 (1999).
- McQueen FM, Stewart N, Crabbe J et al.: Magnetic resonance imaging of the wrist in early rheumatoid arthritis reveals progression of erosions despite clinical improvement. Ann. Rheum. Dis. 58(3), 156–163 (1999).
- Peterfy C: Scratching the surface: articular cartilage disorders in the knee. In: Magnetic Resonance Clinics of North America: Update on the Knee. Rubin D (Ed.), WB Saunders, Philadelphia, PA, USA, 8 (2), 409–430 (2000).
- Forslind K, Larsson EM, Johansson A, Svensson B: Detection of joint pathology by magnetic resonance imaging in patients with early rheumatoid arthritis. Br. J. Rheumatol. 36, 683–688 (1997).
- Østergaard M, Lorenzen I, Henriksen O: Dynamic gadolinium-enhanced MR imaging in active and inactive immunoinflammatory gonarthritis. Acta Radiol. 35, 275–281 (1994).
- Østergaard M, Stoltenberg M, Gideon P: Effects of intraarticular osmic acid on synovial membrane volume and inflammation determined by magnetic resonance imaging. Scand. J. Rheumatol. 24, 5–12 (1995).
- Østergaard M, Stoltenberg M, Gideon P, Sorensen K, Henriksen O, Lorenzen I: Changes in synovial membrane and joint effusion volumes after intraarticular methylprednisolone. Quantitative assessment of inflammatory and destructive changes in arthritis by MRI. J. Rheumatol. 23, 1151–1161 (1996).
- Pirich C, Schwameis E, Bernecker P et al.: Influence of radiation synovectomy on articular cartilage, synovial thickness and enhancement as evidenced by MRI in patients with chronic synovitis. J. Nucl. Med. 40, 1277–1284 (1999).
- Peterfy C, van Dijke C, Lu Y et al.: Quantification of the volume of articular cartilage in the metacarpophalangeal joints of the hand: accuracy and precision of three-dimensional MR imaging. Am. J. Radiol. 165, 371–375 (1995).
- Østergaard M, Peterfy C, Conaghan P et al.: OMERACT rheumatoid arthritis magnetic resonance imaging studies. Core set of MRI acquisitions, joint pathology definitions, and the OMERACT RA-MRI scoring system. J. Rheumatol. 30(6), 1385–1386 (2003).
- Østergaard M, Klarlund M, Lassere M et al.: Interreader agreement in the assessment of magnetic resonance images of rheumatoid arthritis wrist and finger joints – an international multicenter study. J. Rheumatol. 28(5), 1143–1150 (2001).
- Conaghan P, Lassere M, Østergaard M et al.: OMERACT rheumatoid arthritis magnetic resonance imaging studies. Exercise 4: an international multicenter longitudinal study using the RA-MRI Score. J. Rheumatol. 30(6), 1376–1379 (2003).
- Lassere M, McQueen F, Østergaard M et al.: OMERACT rheumatoid arthritis magnetic resonance imaging studies. Exercise 3: an international multicenter reliability study using the RA-MRI Score. J. Rheumatol. 30(6), 1366–1375 (2003).
- Conaghan P, Bird P, Ejbjerg B et al.: The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the metacarpophalangeal joints. Ann. Rheum. Dis. 64(Suppl. 1), I11–I21 (2005).
- Ejbjerg B, McQueen F, Lassere M et al.: The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the wrist joint. Ann. Rheum. Dis. 64(Suppl 1), I23–I47 (2005).
- Bird P, Lassere M, Shnier R, Edmonds J: Computerized measurement of magnetic resonance imaging erosion volumes in patients with rheumatoid arthritis: a comparison with existing magnetic resonance imaging scoring systems and standard clinical outcome measures. Arthritis Rheum. 48(3), 614–624 (2003).
- Døhn UM, Ejbjerg BJ, Hasselquist M et al.: Rheumatoid arthritis bone erosion volumes on CT and MRI: reliability and correlations with erosion scores on CT, MRI and radiography. Ann. Rheum. Dis. 66(10), 1388–1392 (2007).
- Østergaard M, Hansen M, Stoltenberg M et al.: Magnetic resonance imagingdetermined synovial membrane volume as a marker of disease activity and a predictor of progressive joint destruction in the wrists of patients with rheumatoid arthritis. Arthritis Rheum. 42(5), 918–929 (1999).
- Emond PD, Choi A, O’Neill J, Xie J, Adachi R, Gordon CL: The development of EERA: software for assessing rheumatic joint erosions. Can. Assoc. Radiol. J. 60(2), 63–68 (2009).
- Døhn UM, Ejbjerg BJ, Hasselquist M et al.: Detection of bone erosions in rheumatoid arthritis wrist joints with magnetic resonance imaging, computed tomography and radiography. Arthritis Res. Ther. 10(1), R25 (2008).
- Døhn UM, Ejbjerg BJ, Hasselquist M et al.: Rheumatoid arthritis bone erosion volumes on CT and MRI: reliability and correlations with erosion scores on CT, MRI and radiography. Ann. Rheum. Dis. 66(10), 1388–1392 (2007).
- Perry D, Stewart N, Benton N et al.: Detection of erosions in the rheumatoid hand; a comparative study of multidetector computerized tomography versus magnetic resonance scanning. J. Rheumatol. 32(2), 256–267 (2005).
- Savnik A, Malmskov H, Thomsen HS et al.: MRI of the arthritic small joints: comparison of extremity MRI (0.2 T) vs high-field MRI (1.5 T). Eur. Radiol. 11, 1030–1038 (2001).
- Taouli B, Zaim S, Peterfy CG et al.: Rheumatoid arthritis of the hand and wrist: comparison of three imaging techniques. Am. J. Roentgenol. 182, 937–943 (2004).
- Conaghan PG, Ejbjerg B, Lassere M et al.: Multicenter reliability study of extremitymagnetic resonance imaging in the longitudinal evaluation of rheumatoid arthritis. J. Rheumatol. 34(4), 857–858 (2007).
- Bird P, Ejbjerg B, Lassere M et al.: A multireader reliability study comparing conventional high-field magnetic resonance imaging with extremity low-field MRI in rheumatoid arthritis. J. Rheumatol. 34(4), 854–856 (2007).
- Sharp JT, Young DY, Bluhm GB et al.: How many joints in the hands and wrists should be included in a score of radiologic abnormalities used to assess rheumatoid arthritis? Arthritis Rheum. 28(12), 1326–1335 (1985).
- Bird P, Conaghan P, Ejbjerg B et al.: The development of the EULAR-OMERACT rheumatoid arthritis MRI reference image atlas. Ann. Rheum. Dis. 64(Suppl. 1), I8–I10 (2005).
- McQueen F: Magnetic resonance in early inflammatory arthritis: what is its role? Rheumatology 39, 700–706 (2000).
- Conaghan P, Bird P, Ejbjerg B et al.: The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the metacarpophalangeal joints. Ann. Rheum. Dis. 64(Suppl. 1), I11–I21 (2005). & Excellent summary of current metacarpophalangeal joint MRI scoring methods.
- Conaghan P, Bird P, Ejbjerg B et al.: The EULAR-OMERACT rheumatoid arthritis MRI reference image atlas: the metacarpophalangeal joints. Ann. Rheum. Dis. 64(Suppl. 1), I11–I21 (2005).
- Reece R, Kraan MC, Radjenovic A et al.: Comparative assessment of leflunomide and methotrexate for the treatment of rheumatoid arthritis, by dynamic enhanced magnetic resonance imaging. Arthritis Rheum. 46, 366–372 (2002).
- Bird P, Lassere M, Shnier R, Edmonds J. Computerized measurement of magnetic resonance imaging erosion volumes in patients with rheumatoid arthritis: a comparison with existing magnetic resonance imaging scoring systems and standard clinical outcome measures. Arthritis Rheum. 48(3), 614–624 (2003).
- Dohn UM, Ejbjerg B, Hasselquist M et al.: Rheumatoid arthritis bone erosion volumes on CT and MRI: reliability and correlations with erosion scores on CT, MRI and radiography. Ann. Rheum. Dis. 66, 1388-1392 (2007).
- Ejbjerg B, Narvestad E, Jacobsen S, Thomsen HS, Østergaard M: Optimised low cost, low field dedicated extremity MRI is highly specific and sensitive for synovitis and bone erosions in rheumatoid arthritis wrist and finger joints: comparison with high field MRI and radiography. Ann. Rheum. Dis. 64, 1280-1287 (2005).