Editorial - Journal of Agriculture (2023) Volume 6, Issue 4
Silicon Fertilization Enhances the Resistance of Tobacco Plants to Combined Cd and Pb Contamination: Physiological and Microbial Mechanisms
Aaron Shew*
Department of Biology, Lund University, Ecology Building, Sweden
Department of Biology, Lund University, Ecology Building, Sweden
E-mail: Aaronshwen4@gmail.com
Received: 01-Aug-2023, Manuscript No. jagri-23-108485; Editor assigned: 03-Aug-2023, Pre-QC No. jagri-23-108485 (PQ); Reviewed: 17- Aug-2023, QC No. jagri-23-108485; Revised: 22-Aug-2023, Manuscript No. jagri-23-108485 (R); Published: 29-Aug-2023; DOI: 10.37532/ jagri.2023.6(4).97-99
Abstract
Environmental pollution due to heavy metal contamination, such as cadmium (Cd) and lead (Pb), has become a significant concern worldwide. These toxic metals not only threaten ecosystem health but also pose risks to human health through the food chain. In recent years, researchers have explored various approaches to mitigate heavy metal-induced toxicity in plants. Among these strategies, silicon (Si) fertilization has gained attention for its potential to enhance plant resistance to metal stress. This article examines the effects of silicon fertilization on tobacco plants subjected to combined Cd and Pb contamination, elucidating the physiological and microbial mechanisms involved in conferring resistance
Keywords
Environmental pollution • Heavy metal-induced toxicity • Silicon fertilization • Plant resistance • Physiological and microbial mechanisms
Introduction
Cadmium and lead are common heavy metals found in contaminated soils, primarily originating from industrial activities, mining, and agricultural practices. Once absorbed by plants, these metals can negatively affect growth, development, and yield. The use of silicon fertilization as a means to counteract metal toxicity in plants has shown promising results in previous studies. Silicon, an essential nutrient for plants, has been reported to improve the stress tolerance of various crops, including rice, wheat, and barley. However, its effects on tobacco plants exposed to both Cd and Pb contamination remain relatively unexplored [1, 2].
Physiological mechanisms
Silicon’s protective role against heavy metal stress in tobacco plants involves several physiological mechanisms. Firstly, it enhances the plant’s antioxidant defense system, including the production of enzymes such as superoxide dismutase (SOD), catalase (CAT), and peroxidase (POD) [3]. These enzymes scavenge harmful reactive oxygen species (ROS) generated by heavy metal exposure, reducing oxidative damage to cellular components. Secondly, silicon improves the regulation of mineral nutrients, such as calcium (Ca) and potassium (K), thereby reducing the uptake and translocation of Cd and Pb within the plant. Thirdly, Si deposition in the cell walls forms a physical barrier, limiting the entry and transport of toxic metals from roots to shoots [4].
Analysis of plant and soil
Growth parameters and biomass: The growth parameters of tobacco (plant height, leaf length, leaf width, leaf area) were measured before harvest. After harvesting, the tobacco tissues (root, stem and leaf ) were separated and washed manually, and the biomass was determined by weighing after drying at 75 ± 5 ◦C. Each treatment had three replications [5].
Microbial mechanisms
The beneficial effects of silicon on tobacco plants under heavy metal stress are not limited to its direct interactions with the plant. Silicon also influences the rhizosphere microbial community and its activities. Increased silicon availability promotes the growth and activity of beneficial microorganisms, such as mycorrhizal fungi and plant growth-promoting bacteria. These microbes play crucial roles in enhancing plant nutrient uptake and reducing heavy metal bioavailability in the soil. Additionally, some microbial species possess the ability to immobilize or detoxify heavy metals, further aiding in the protection of tobacco plants against Cd and Pb contamination [6,7].
Discussion
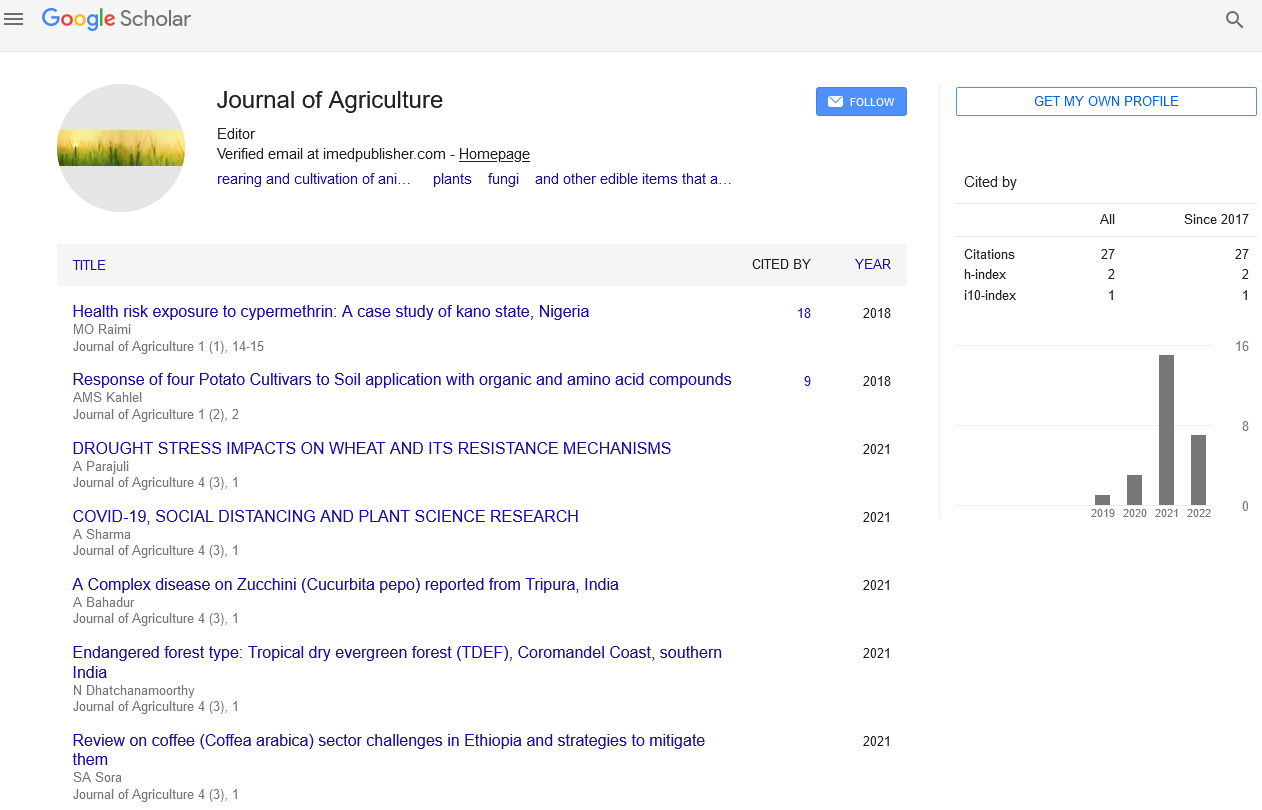
Amendment of soils with Si fertilizer has been suggested as a potential remediation strategy to relieve heavy metal stress, because Si could stimulate plant growth and strengthen plant tolerance against abiotic and biotic stresses [8]. We found that Si fertilization reduced the Cd content of tobacco plants by 42.0–55.5 % and Pb content by 17.2–25.6 %, compared to the control (Fig. 1). Both OSiF and MSiF can form precipitates with heavy metals, thereby hindering the transportation of Cd and Pb within soils Consistent with this, we found that Si fertilizer addition reduced concentrations of available Cd and Pb in soils under Cd-Pb stress (Table 1). Though the Cd-Pb stress resulted in Cd and Pb enrichment in the tobacco plants, we found that both OSiF and MSiF decreased the heavy metal content in the root, stem and leaves under Cd-Pb stress [9]. (2019a) also found that both organic and mineral Si fertilizers decreased Cd and Pb content of various tissues of wheat, as Si fertilizer hindered the transfer of Cd and Pb by forming precipitates with Cd and Pb. In addition, Si addition was found to ameliorate the negative effect of Cd and Cu heavy metals on root function in a study of rice plants (Kim et al., 2014), while another study found that the application of nano-Si effectively facilitated the transport of mineral nutrients, increasing nutrient content of plant tissues [10].
Conclusion
Silicon fertilization represents a promising eco-friendly strategy to enhance the resistance of tobacco plants to combined Cd and Pb contamination. Through a combination of physiological and microbial mechanisms, silicon mitigates heavy metal-induced oxidative stress, regulates nutrient uptake, and modifies the rhizosphere microbial community. As a result, tobacco plants treated with silicon exhibit improved growth, reduced metal accumulation, and enhanced overall resilience to metal stress. This research underscores the potential of silicon as a sustainable and cost-effective approach for phytoremediation and plant protection in heavy metal-contaminated environments. However, further investigations are necessary to fully understand the underlying molecular and genetic processes involved in silicon-mediated metal tolerance in tobacco and other plant species. This study illustrated that amendment of soils with Si fertilizer could mitigate the toxic influence of Cd and Pb on tobacco plants, resulting in higher plant height, leaf length, leaf width, and leaf area. Simultaneously, Si fertilizer amendment decreased Cd and Pb contents in the soil and reduced concentrations in plant tissues. The potential mechanisms for relieving Cd and Pb stresses by Si fertilizer include a reduced effectiveness of heavy metals due to forming precipitates with Si, enhanced antioxidant defenses and an increased photosynthetic capacity. We also found that the bacterial phyla Proteobacteria and Acid bacteria exhibited a high tolerance to the heavy metals, and Si fertilizer improved stress resistance via changes in microbial community composition, by favoring microorganisms with metabolic pathways linked to heavy metal resistance. Application of organic and mineral Si fertilizers could therefore offer a viable strategy for reducing Cd-Pb stress in tobacco-planted soils, and the utility for militating against combined Cd-Pb stress should be further tested for other plant types.
References
- Naylor, Rosamond L. Oil crops, aquaculture, and the rising role of demand: A fresh perspective on food security.Global Food Securit.11,17-25(2016).
- Khan G, Haleem AA. Plant Physiological Responses to Engineered Nanoparticles. ENPS.12 (1),85-99(2021).
- Ask EI, Azanza RV. Advances in cultivation technology of commercial eucheumatoid species: a review with suggestions for future research.Aquac.206,257-277(2002).
- Zabrieski Z, Morrell E, Hortin J et al. Pesticidal activity of metal oxide nanoparticles on plant pathogenic isolates of Pythium.Ecotoxicology.24 (6), 1305-1314(2015).
- Shah J, Ali A. Combined effect of Bacillus fortis IAGS 223 and zinc oxide nanoparticles to alleviate cadmium phytotoxicity in Cucumis melo. Plant PhysiolBiochem. 158, 1-12(2021).
- Reverter M, Sarter S, Caruso D et al. Aquaculture at the crossroads of global warming and antimicrobial resistance. NatCommun.11(1), 1870-1879 (2020).
- Ogello EO, Mlingi FT, Nyonje BM et al. Can integrated livestock-fish culture be a solution to East Afircan's food insecurity? A Review.African J Food Agric Nutr Dev.13 (4), 8058-8078 (2013).
- Nesar A, Thompson S, Giovanni M. Organic aquaculture productivity, environmental sustainability, and food security: insights from organic agriculture.Food Security.12, 1253-1267 (2020).
- Jennings L, Simon M. Aquatic food security: insights into challenges and solutions from an analysis of interactions between fisheries, aquaculture, food safety, human health, fish and human welfare, economy and environment.Fish Fish. 17(4), 893-938 (2016).
- Kiran P, Adi J. Aquaculture role in global food security with nutritional value: a review.TranslAnimSci. 3(2), 903-910 (2019).
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref
Indexed at, Google Scholar, Crossref