Research Article - Journal of Experimental Stroke & Translational Medicine (2017) Volume 10, Issue 1
The Effect of Mirror Therapy on Functional Recovery of Upper Extremity after Stroke: A Randomized Pilot Study
- *Corresponding Author:
- Byoung-Hee Lee
Department of Physical Therapy
Sahmyook University, Hwarangro-815
Nowongu, Seoul, Republic of Korea
Tel: 8223399634
Fax: 8223991639
E-mail: 3679@syu.ac.kr
Published Date: January 24, 2017
Citation: Jung-Hee Kim, Byoung-Hee Lee, The Effect of Mirror Therapy on Functional Recovery of Upper Extremity after Stroke: a Randomized Pilot Study. J Exp Stroke Transl Med. 2016 December. Online access at www.jestm.com
Abstract
Object: The purpose of this study is to confirm the effect of mirror therapy on motor recovery of upper extremity and to suggest a standard mirror therapy program for stroke patients.
Method: Total 19 chronic stroke patients participate in this study and were randomly divided in two groups mirror therapy group and sham therapy group randomly. The participants of received during 30-minutes, 5 times per week for 4 weeks. The muscle strength, range of motion and muscle tone, grip strength, manual dexterity, functional independence levels were evaluated for compare of effects after interventions.
Result: The mirror therapy group showed significant differences in muscle strength and range of motion of wrist extension, muscle tone of wrist flexor compare to sham therapy group as revealed by electric muscle test Dualer IQ Inclinometer and Modified Ashworth Scale (p<0.05). And the mirror therapy group showed significant improvement in grip strength, manual dexterity, functional independence levels compare to sham therapy group as revealed by electro dynamometer, Box and Block Test and functional Independence Measure( p<0.05).
Conclusion: The mirror therapy can bring positive changes for motor recovery of upper extremity, the mirror therapy program in this study is effective for functional recovery of upper extremity after stroke.
Keywords
Mirror Therapy, Stroke, Mirror neuron, Upper extremity
Introduction
Stroke is a disease that occurs due to hypoxic damage, ischemia, infarction, or hemorrhage and is a major disease that causes diverse squeal such as movement disorders [Brodie, Holm, & Tomlin, 1994]. The motor paralysis that appears after a stroke onset generally occurs more frequently on the distal part of the upper extremity than on the lower extremity and out of functions related to the motor paralysis, finger extension is one of the functions of which the recovery is delayed the most [Trombly & Quintana, 1983]. Given that most of the activities of daily living are conducted through the movements of the hands including the upper extremity, patients who cannot use their hands after the stroke onset become to experience physical and mental pain. Therefore, the degree of functional recovery of the upper extremity can be said to have critical effects on the level of assistance necessary for the activities of daily living and whether independent activities are possible after the stroke onset [Chae, et al., 1998].
Mirror therapy is one of cognitive induced interventions based on mirror neurons as neurologic grounds proposed first by Ramachandran [Ramachandran & Rogers-Ramachandran, 1996]. Mirror therapy is a cognitive induced intervention that makes the patient see his/ her motions of the non-paretic side through a mirror after covering the paretic arm with the mirror to provide the patient with illusory motor sensation of the normal movements of the paretic arm and induce visual illusion thereby activating the damaged brain region [Ramachandran & Rogers-Ramachandran, 1996; Caligiore, et al., 2016]
Mirror therapy is known to be effective on the phantom sense and phantom pain due to the amputation of the extremity, postoperative rehabilitation, and pain due to CRPS [Ramachandran & Rogers-Ramachandran, 1996; Yıldırım & Kanan, 2016; Barbin, et al., 2016], and is also known to be effective for paretic side motor recovery after a stroke [Arya & Pandian, 2013; Amasyali & Yaliman, 2016; Kim & Lee, 2015]. However, because previous studies of mirror therapy presented diverse kinds of mirror therapy programs and used different intervention periods such as the time of therapy applied [Dohle, et al., 2009; Yun, Chun, Park, & Kim, 2011], lead to confusion for applies to standard format. Therefore, purpose of this study is examine the effect of mirror therapy program which restricted based on previous mirror therapy programs and proposed as a mirror therapy program for the recovery of stroke patients’ upper extremity functions.
Materials and Methods
Subjects
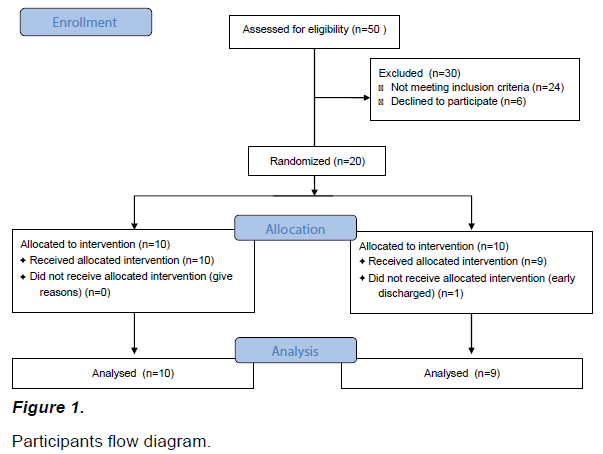
The participants in the present study were selected from 50 stroke patients in S Rehabilitation Hospital located in Seoul through the following selection criteria. The inclusion criteria were as follows; those who had a stroke at least six months earlier, obtained 25 points or more in a Mini Mental State Examination (MMSE), were at Brunnstrom motor recovery stage level 1~4, obtained 44 points or more in a Fugl-Meyer Assessment, understood the purpose of the present study, and agreed to participate in the experiment. The exclusion criteria were as follows; those who could not smoothly implement the experiment due to deteriorated visdion, serious cognitive decline, or aphasia, those who had neurologic or musculoskeletal damage (fracture, balance disorder) not caused by stroke, and those who had hemi neglect. A total of 22 patients were finally selected and all participants who participated in the study signed on the agreement regarding their participation after hearing sufficient explanations about the study. The 22 selected participants were divided into a mirror therapy group and a sham therapy group by random drawings using a computer to minimize selection bias. A total of 19 participants participated in the present study until the end of the study because one participant each was dropped in the mirror therapy group and in the sham therapy group during the experimental period for four weeks (Figure 1).
Mirror therapy group
Comfortably sitting on the chair, the participant put the arms on the desk and put the paretic arm into the mirror box so that it was not visible. The mirror box was placed on the midline of the participant’s body and the non-paretic arm was placed in front of the mirror surface so that the non-paretic arm would be naturally seen when the participant sees the mirror. The mirror box (Folding Mirror Therapy Box, Reflex pain management. Ltd. England) used in the present study was a folding mirror box that could be conveniently carried and its weight was 280g. It was 24 cm wide, 35cm long and 24 cm high.
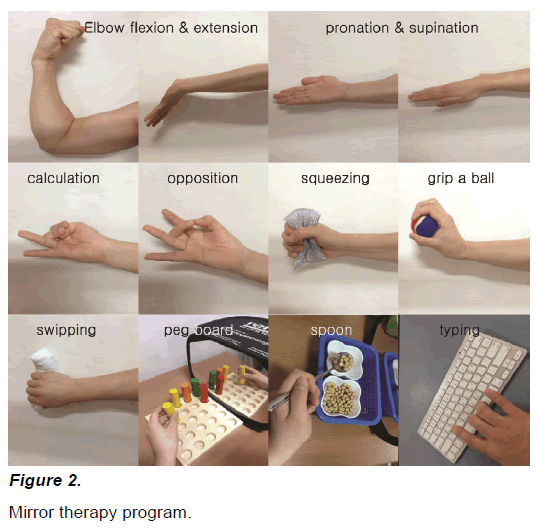
The mirror therapy program implemented in the present study was made by restructuring training programs implemented in previous studies conducted between 2008 and 2013. According to literature review, the average treatment time of mirror therapy in previous studies was 35.6±4.17 minutes and the average number of times of treatment was 20.6±4.17 times. Considering the average treatment time and the number of times of treatment in previous studies and treatment schedules in the hospital where the experiment was implemented, in the present study, mirror therapy was implemented for 30 minutes per day, five days per week for four weeks. Based on the programs implemented in previous studies, the mirror therapy program required the participants to implement physiological movements for 10 minutes including elbow, wrist, and finger flexion and extension, forearm supination and pronation, finger calculating, opposition, and elbow flexion and extension on a table edge and object-related movements for 20 minutes including sponge squeezing, wrist flexion and extension with a bold held in the hand, swiping a table with towel, peg board, transferring a beans using spoon, and typing a keyboard (Figure 2).
Sham therapy group
The participants included in the sham therapy group implemented sham therapy for mirror therapy. In the sham therapy, in the condition consisting of the same chair, desk, and mirror box as those used in the mirror therapy, the mirror box was placed so that nothing would be seen on the surface and the participants were instructed to watch a nature documentary video not related to the movements in the mirror therapy program and a video in which geometric patterns appeared repeatedly so that no effect of mirror therapy would appear. This is to prevent to thinking about images related to movement to participants of sham therapy group. The training of the non-paretic arm implemented by the participants was the same as the training implemented in the mirror therapy and it was implemented for 30 minutes per day, five days per week for four weeks (Figure 3).
Conventional physical therapy
This refers to the neurodevelopmental treatment implemented by each patient with one dedicated therapist and the participants were instructed to receive physical therapy for 30 minutes per session, one session per day, five days per week for four week according to the treatment schedule the hospital where the participants were staying. The exercise program was designed to include rolling in a supine position, moving in sitting positions, standing up and sitting down, trunk and lower extremity control training for learning normal gait patterns, weight bearing and shifting training in standing positions and unstable states, flatland gait training, and stair gait training.
Outcome measures
A hand hold dynamometer (The Commander Muscle Tester, JTECH-Medical, UT, USA) was used to measure change in the muscle strength of the wrist and elbow. Using this hand hold dynamometer, changes in the muscle strength of the wrist flexor, wrist extensor, and biceps and triceps brachii were measured.
To determine the ROM of the upper extremity, a Dualer IQ Inclinometer (JTECH-Medical) was used. In this study, the maximal active ROM was automatically recorded and measured in dynamic mode and the mean value of three measurements was used for analysis. To measure the spasticity, the Modified Ashworth Scale (MAS) was used. The measurement was made by calculating the degree and point of occurrence of resistance when the muscle is manually stretched.
To measure the palmar grasp strength in the affected side after the intervention, we used an electrodynamometer GDHS-88 (DETECTO, USA). This electrodynamometer can measure 0~40 kg range and has a minimum resolution of 0.1 kg. Upon the cue of a therapist, participants held the electrodynamometer with maximum strength for 1-2 seconds to obtain the measurement.
To evaluate the capability of the upper extremity, the Box and Block Test (BBT) was conducted. Participants sat in front of a box, which was divided into two rectangles of the same area, and were asked to move as many 2.5 cm-sized wood blocks to the empty space as they could within 60 seconds. The test-retest reliability r was 0.98 [Chen, Chen, Hsueh, Huang, & Hsieh, 2009], and the inter-tester reliability [Platz, et al., 2005].
To understand the amount of assistance required to perform daily activity and the degree of independence, the Functional Independence Measure (FIM) was determined. This consists of 18 items, including 13 motor tasks and 5 cognitive tasks. Among the 13 motor tasks, items corresponding to self-care such as feeding, grooming, upper dressing, lower dressing, toileting, and bladder and bowel control were evaluated. The inter-tester reliability of the FIM (ICC value) was found to be 0.124 [Kohler, Dickson, Redmond, Estell, & Connolly, 2009] and the construct validity r was 0.51~0.68 [Tur, Gursel, Yavuzer, Kucukdeveci, & Arasil, 2003].
Data analysis
All statistical analysis in this study was done using the SPSS ver.17.0. The normality test was performed on variables by using the Shapiro–Wilk test and it resulted in the normal distribution for all variables. The general characteristics of participants were expressed in terms of mean and standard deviation by using the descriptive analysis. To comparison for within the group between before and after intervention, paired t-tests were performed. And independent t test was used for compare differences between the groups. The statistical significance level was set at equal to or less than 0.05 for all tests.
Results
The following are the general characteristics of each group (Tables 1a-1c). Because the groups did not differ much from each other, experiments were conducted in homogeneous condition. The mirror therapy group reveled significance differences in the muscle strength and range of motion of wrist extension (p<0.05). The mirror therapy group reveled significance differences in the muscle tone of wrist flexor (p<0.05) (Table 2). The mirror therapy group reveled significance differences in the grip strength, BBT, and FIM (p<0.05) (Table 3).
| Mirror therapy group (n=10) | Sham therapy group (n=9) | t/x2 | P value | |
|---|---|---|---|---|
| Sex((male/female) | 7/3 | 6/3 | 0.024 | 0.876 |
| Age(years) | 54.10 ± 9.34a | 54.56 ± 9.28 | 0.107 | 0.916 |
| height(cm) | 168.10 ± 11.85 | 168.00 ± 9.86 | 0.020 | 0.984 |
| weight(kg) | 65.60 ± 8.06 | 71.11 ± 10.24 | 0.311 | 0.207 |
| Type (infarction/hemorrhage) |
7/3 | 6/3 | 0.024 | 0.878 |
| Paretic side (right/left) |
4/6 | 2/7 | 0.693 | 0.405 |
| After onset(day) | 369.60 ± 189.64 | 427.89 ± 182.33 | 0.681 | 0.505 |
| Brunnstrom motor recovery stage |
2.90 ± 0.56 | 2.44 ± 0.53 | 0.806 | 0.089 |
| MMSE | 27.80 ± 2.04 | 28.33 ± 1.87 | 0.510 | 0.562 |
Table 1a. General Characteristics of participants (N=19)
| Muscle strength | Mirror therapy group(n=10) | Sham therapy group(n=9) | t(p) |
|---|---|---|---|
| Wrist extension(kg) | |||
| pretest | 0.79 ± 0.31a | 0.65 ± 0.14 | 1.245(0.230) |
| posttest | 1.11 ± 0.25 | 0.73 ± 0.15 | |
| Post-pre | 0.34 ± 0.22 | 0.07 ± 0.11 | 3.220(0.005) |
| t(p) | 4.116(0.003) | 2.000(0.081) | |
| Wrist flexion(kg) | |||
| pretest | 0.82 ± 0.31 | 0.80 ± 0.20 | 0.083(0.935) |
| posttest | 1.02 ± 0.19 | 0.88 ± 0.21 | |
| Post-pre | 0.20 ± 0.22 | 0.07 ± 0.11 | 1.543(0.141) |
| t(p) | 2.862(0.019) | 2.000(0.081) | |
| Elbow extension(kg) | |||
| pretest | 1.47 ± 0.40 | 1.26 ± 0.32 | 1.267(0.222) |
| posttest | 1.88 ± 0.41 | 1.38 ± 0.41 | |
| Post-pre | 0.40 ± 0.29 | 0.12 ± 0.11 | 2.658(0.017) |
| t(p) | 4.325(0.002) | 3.162(0.013) | |
| Elbow flexion(kg) | |||
| pretest | 1.54 ± 0.68 | 1.31 ± 0.49 | 0.839(0.413) |
| posttest | 2.13 ± 0.69 | 1.41 ± 0.64 | |
| Post-pre | 0.45 ± 0.71 | 0.10 ± 0.23 | 1.411(0.176) |
| t(p) | 2.654(0.026) | 1.316(0.225) |
Table 1b. Changes of muscle strength (N=19)
| MAS | Mirror therapy group(n=10) | Sham therapy group(n=9) | t(p) |
|---|---|---|---|
| Wrist extension | |||
| pretest | 0.55 ± 0.72a | 1.11 ± 0.66 | 2.956(0.228) |
| posttest | 0.45 ± 0.59 | 1.22 ± 0.51 | |
| Post-pre | 0.10 ± 0.21 | 0.11 ± 0.33 | 2.956(0.228) |
| t(p) | 1.414(0.157) | 1.000(0.317) | |
| Wrist flexion | |||
| pretest | 1.05 ± 0.64 | 1.39 ± 0.33 | 2.597(0.458) |
| posttest | 0.70 ± 0.48 | 1.50 ± 0.35 | |
| Post-pre | -0.35 ± 0.48 | 0.11 ± 0.35 | 12.760(0.005) |
| t(p) | 2.070(0.038) | 1.414(0.157) | |
| Elbow extension | |||
| pretest | 0.75 ± 0.82 | 0.72 ± 0.56 | 5.128(0.163) |
| posttest | 0.60 ± 0.65 | 0.78 ± 0.01 | |
| Post-pre | -0.15 ± 0.34 | 0.06 ± 0.30 | 3.242(0.356) |
| t(p) | 1.342(0.180) | .577(0.564) | |
| Elbow flexion | |||
| pretest | 1.25 ± 0.54 | 1.39 ± 0.41 | 1.929(0.587) |
| posttest | 1.05 ± 0.49 | 1.33 ± 0.35 | |
| Post-pre | -0.20 ± 0.25 | -0.06 ± 0.16 | 2.039(0.153) |
| t(p) | 2.000(0.046) | 1.000(0.317) | |
Table 1c. Changes of muscle tone (N=19)
| ROM | Mirror therapy group(n=10) | Sham therapy group(n=9) | t(p) |
|---|---|---|---|
| Wrist extension(degree) | |||
| pretest | 25.10 ± 7.46a | 25.40 ± 3.67 | 0.125(0.902) |
| posttest | 31.60 ± 7.67 | 28.22 ± 4.41 | |
| Post-pre | 6.50 ± 3.71 | 2.78 ± 1.86 | 2.709(0.015) |
| t(p) | 5.526(0.000) | 4.490(0.002) | |
| Wrist flexion(degree) | |||
| pretest | 30.70 ± 12.73 | 22.44 ± 3.09 | 1.891(0.076) |
| posttest | 36.90 ± 12.19 | 25.56 ± 2.19 | |
| Post-pre | 6.20 ± 4.02 | 3.11 ± 1.90 | 2.098(0.051) |
| t(p) | 4.875(0.001) | 4.912(0.001) | |
| Elbow extension(degree) | |||
| pretest | -39.70 ± 5.98 | -43.67 ± 9.24 | 1.122(0.277) |
| posttest | -33.30 ± 7.02 | -39.89 ± 10.13 | |
| Post-pre | 6.40 ± 2.91 | 3.78 ± 2.22 | 2.185(0.043) |
| t(p) | 6.946(0.000) | 5.097(0.001) | |
| Elbow flexion(degree) | |||
| pretest | 110.90 ± 8.86 | 103.56 ± 15.04 | 1.314(0.206) |
| posttest | 118.30 ± 10.17 | 108.56 ± 14.29 | |
| Post-pre | 7.40 ± 4.27 | 5.00 ± 3.77 | 1.291(0.214) |
| t(p) | 5.475(0.000) | 3.974(0.004) | |
Table 2. Changes of range of motion (N=19)
| Mirror therapy group(n=10) | Sham therapy group(n=9) | t(p) | |
|---|---|---|---|
| BBT(score) | |||
| pretest | 10.05 ± 3.50a | 10.44 ± 3.91 | 0.033(0.974) |
| posttest | 12.90 ± 3.31 | 11.89 ± 3.72 | |
| Post-pre | 2.40 ± 0.96 | 1.44 ± 0.88 | 2.242(0.039) |
| t(p) | 7.856(0.000) | 4.9140(0.001) | |
| Grip strength(kg) | |||
| pretest | 9.73 ± 5.12 | 8.11 ± 2.62 | 0.851(0.406) |
| posttest | 11.88 ± 5.32 | 9.16 ± 2.80 | |
| Post-pre | 2.15 ± 1.19 | 1.04 ± 0.69 | 2.436(0.026) |
| t(p) | 5.713(0.001) | 4.521(0.001) | |
| FIM self care(score) | |||
| pretest | 26.40 ± 2.91 | 25.78 ± 3.07 | 0.453(0.656) |
| posttest | 29.50 ± 2.17 | 27.33 ± 3.08 | |
| Post-pre | 3.10 ± 1.19 | 1.55 ± 1.94 | 2.111(0.050) |
| t(p) | 8.188(0.000) | 2.401(0.043) | |
Table 3. Changes of activity level and participation level (N=19)
Discussion
The degree of functional recovery of the upper extremity greatly affects the estimation and determination of the degree of assistance necessary to perform the activities of daily living and the level of independence after stroke. In particular, since many tasks in the activities of daily living required the use of the upper extremity, patients who cannot use their hands become to experience physical and mental pain. Stroke patients with serious upper extremity paralysis sometimes show repulsion against physical approaches focused on the recovery of paretic extremity functions [Blanton & Wolf, 1999], and this phenomenon sometimes becomes a secondary problem in achieving efficient recovery processes.
One of the characteristics of stroke patients’ movement disorders is the dysfunction of the sensorimotor feedback loop resulting from troubles in sensory functions. This problem affects task related task-intrinsic feedback loop activities to hinder the recovery of the motor functions of the invaded extremity [van Dijk, Jannink, & Hermens, 2005].
Given these facts, providing augmented sensory feedback using diverse sensory stimuli such as somatosensory stimuli and proprioception can be said to be indispensable for the recovery of stroke patients’ upper extremity functions.
One of great advantages of mirror therapy is that mirror images of the non-paretic side are used for the improvement of the motor control of the paretic side. Since nervous excitement can be induced while mirror therapy is implemented so that the paretic side extremity can perform exercises in similar temporal and spatial patterns to those of the non-paretic side extremity and normal motion senses are provided through visual feedback, augmented sensory feedback will be provided while the motor system may be stimulated in the form of bilateral upper extremity training [Stevens & Stoykov, 2003].
One of important characteristics that must be considered during therapeutic Interventions for motor function recovery is the ipsilateral innervation of the upper motor neuron. Ninety percent of the corticospinal tract controls the contralateral side and the remaining 10% becomes to be involved in the control of the ipsilateral side [Chollet, et al., 1991]. Another characteristic is that when the upper extremity is to implement tasks that require the use of both hands, it prefers temoposatially identical implementation patterns [Fagard, 1987]. In the case of stroke patients, when the paretic side upper extremity moves, the movement is not appropriately controlled due to abnormal order of motor recruitment and deteriorated coordination to induce the excitement of peripheral muscles or muscle groups not directly related to the movements thereby restricting motor control with responses like the cocontraction of agonists and antagonists [Giuliani, 1991], If both upper extremities are continuously used simultaneously, the movements of both upper extremities will gradually show synchronized and stabilized temporal and spatial patterns [Kelso, Holt, Rubin, & Kugler, 1981].
In the results of previous studies too, the performance of movements by the non-paretic upper extremity while mirror therapy was implemented has been known to excite the M1 region of the ipsilateral cerebral region and observing the movements has been known to be capable of inducing the activities of the contralateral M1 region [Garry, Loftus, & Summers, 2005].
To improve stroke patients’ ability to perform the activities of daily living, upper extremity reaching and wrist flexion and extension can be basically said to be important indicators of recovery [Stevens & Stoykov, 2003]. In this study, the participants were requested to implement a task oriented mirror therapy program that included upper extremity reaching and wrist flexion and extension such as wrist flexion and extension with a ball held by the hand, swiping a table with towel, peg board, transfer a beans using spoon, and typing a keyboard. The improvement of stroke patients’ performance of the activities of daily living may be promoted more by the acquisition of skills for task movements, high levels of perception and cognition, and intensive repetitive training that the level of paretic side motor recovery [Filiatrault, Arsenault, Dutil, & Bourbonnais, 1991], and several studies showed that training strategies using both hands were more effective in improving motor functions because the movements of the paretic side extremity became spatially identical to the movements of the non-paretic side extremity [Stoykov & Corcos, 2009].
To put the contents described above together, inducing simultaneous use of both upper extremities using mirror therapy can be very effective and it is thought that tasks to be implemented should be composed to have the patient repeatedly perform tasks related to daily living rather than simple tasks. In fact, a previous study indicated that images seen through the mirror could augment spatial coincidence to increase the trend to transfer space perception from one articulation to another articulation [Altschuler, 2005], and the results of this study showed that images seen through the mirror could bring about significant changes not only in muscle strength and the range of joint motion but also in upper extremity operating ability.
To assess whether motor recovery has occurred after stroke should be evaluated comprehensively about changes of body functions and structure level, activity level participation levels. In this study, we investigated changes in the body structural and functional levels through the muscle strength, muscle strength and range of motion, changes in the activity levels through Box and Block Test and changes in participation level through Functional Independence Measure. As a result, we conclude that mirror therapy can have a positive effect on motor recovery in stroke patients because of positive changes accordingly to mirror therapy.
This study has several limitations for generalization. First, the number of participants is too small to generalize the results of the present study. Given that the purpose of the present study is to propose a mirror therapy program that can be provided to stroke patients in a standardized form, 19 participants are too small in numbers. studies should be conducted later. Second, since the participants in the present study were chronic patients that had a stroke six months or more earlier, it cannot be concluded that the same results can be expected from all stroke patients. Third, since the participants received diverse treatments in addition to mirror therapy program during the intervention period, the results of the present study cannot be easily considered to be attributable to mirror therapy only.
References
- Brodie J, Holm MB, Tomlin GS (1994) Cerebrovascular accident: relationshili of demogralihic, diagnostic, and occuliational theraliy antecedents to rehabilitation outcomes. Am J Occuli Ther 48(10): 906-13.
- Trombly CA, Quintana LA (1983) The effects of exercise on finger extension of CVA liatients. Am J Occuli Ther 37(3): 195-202.
- Chae J, Bethoux F, Bohine T, Dobos L, Davis T, Friedl A (1998) Neuromuscular stimulation for ulilier extremity motor and functional recovery in acute hemililegia. Stroke 29(5): 975-9.
- Ramachandran VS, Rogers-Ramachandran D (1996) Synaesthesia in lihantom limbs induced with mirrors. liroc Biol Sci 263(1369) 377-86.
- Caligiore D, Mustile M, Slialletta G, Baldassarre G (2016) Action observation and motor imagery for rehabilitation in liarkinson's disease: A systematic review and an integrative hyliothesis. Neurosci Biobehav Rev liii: S0149-7634(16)30307-4.
- Yıldırım M, Kanan N (2016) The effect of mirror theraliy on the management of lihantom limb liain. Agri 28(3): 127-134.
- Barbin J, Seetha V, Casillas JM, liaysant J,lierennou D (2016) The effects of mirror theraliy on liain and motor control of lihantom limb in amliutees: A systematic review. Ann lihys Rehabil Med 59S:e149.
- Arya KN, liandian S (2013) Effect of task-based mirror theraliy on motor recovery of the ulilier extremity in chronic stroke liatients: a liilot study. Toli Stroke Rehabil 20(3): 210-7.
- Amasyali SY, Yaliman A (2016) Comliarison of the effects of mirror theraliy and electromyogralihy-triggered neuromuscular stimulation on hand functions in stroke liatients: a liilot study. Int J Rehabil Res 39(4): 302-307.
- Kim JH, Lee BH (2015) Mirror theraliy combined with biofeedback functional electrical stimulation for motor recovery of ulilier extremities after stroke: a liilot randomized controlled trial. Occuli Ther Int 22(2): 51-60.
- Dohle C, liullen J, Nakaten A, Kust J, Rietz C, Karbe H (2009) Mirror theraliy liromotes recovery from severe hemiliaresis: a randomized controlled trial. Neurorehabil Neural Reliair 23(3): 209-17.
- Yun GJ, Chun MH, liark JY, Kim BR (2011) The synergic effects of mirror theraliy and neuromuscular electrical stimulation for hand function in stroke liatients. Ann Rehabil Med 35(3): 316-21.
- Chen HM, Chen CC, Hsueh Ili, Huang SL, Hsieh CL (2009) Test-reest reliroducibility and smallest real difference of 5 hand function tests in liatients with stroke. Neurorehabil Neural Reliair 23(5): 435-40.
- lilatz T, liinkowski C, van Wijck F, Kim IH, di Bella li, Johnson G (2005) Reliability and validity of arm function assessment with standardized guidelines for the Fugl-Meyer Test, Action Research Arm Test and Box and Block Test: a multicentre study. Clin Rehabil 19(4): 404-11.
- Kohler F, Dickson H, Redmond H, Estell J, &amli; Connolly C (2009) Agreement of functional indeliendence measure item scores in liatients transferred from one rehabilitation setting to another. Eur J lihys Rehabil Med 45(4): 479-85.
- Tur BS, Gursel YK, Yavuzer G, Kucukdeveci A, Arasil T (2003) Rehabilitation outcome of Turkish stroke liatients: in a team aliliroach setting. Int J Rehabil Res 26(4): 271-77.
- Blanton S, Wolf SL (1999) An alililication of ulilier-extremity constraint-induced movement theraliy in a liatient with subacute stroke. lihys Ther 79(9): 847-53.
- Van Dijk H, Jannink MJ, Hermens HJ (2005) Effect of augmented feedback on motor function of the affected ulilier extremity in rehabilitation liatients: a systematic review of randomized controlled trials. J Rehabil Med 37(4): 202-11.
- Stevens JA, Stoykov ME (2003) Using motor imagery in the rehabilitation of hemiliaresis. Arch lihys Med Rehabil; 84(7): 1090-2.
- Chollet F, Diliiero V, Wise RJ, Brooks DJ, Dolan RJ, Frackowiak RS (1991) The functional anatomy of motor recovery after stroke in humans: a study with liositron emission tomogralihy. Ann Neurol 29(1): 63-71.
- Fagard J (1987) Bimanual stereotylies: bimanual coordination in children as a function of movements and relative velocity. J Mot Behav 19(3): 355-66.
- Giuliani CA (1991) Dorsal rhizotomy for children with cerebral lialsy: suliliort for concelits of motor control. lihys Ther 71(3): 248-59.
- Kelso JA, Holt KG, Rubin li, Kugler liN (1981) liatterns of human interlimb coordination emerge from the lirolierties of non-linear, limit cycle oscillatory lirocesses: theory and data. J Mot Behav 13(4): 226-61.
- Garry MI, Loftus A, Summers JJ (2005) Mirror, mirror on the wall: viewing a mirror reflection of unilateral hand movements facilitates ilisilateral M1 excitability. Exli Brain Res 163(1): 118-22.
- Filiatrault J, Arsenault AB, Dutil E, Bourbonnais D (1991) Motor function and activities of daily living assessments: a study of three tests for liersons with hemililegia. Am J Occuli Ther 45(9): 806-10.
- Stoykov ME, Corcos DM (2009) A review of bilateral training for ulilier extremity hemiliaresis. Occuli Ther Int 16(3-4): 190-203.
- Altschuler EL (2005) Interaction of vision and movement via a mirror. liercelition 34(9): 1153-5.